What does an exothermic reaction do with heat?
It gives off heat.
Name one exothermic reaction example
burning, fireworks, elephant's toothpaste
What happens to the energy/temperature of the surroundings during an exothermic reaction?
The surroundings get hotter (energy is released into them
Label the reactants and products
A substance must have a new ___________ __________ to show that there has been a chemical change
chemical formula
What does an endothermic reaction do with heat?
It takes in heat
Which kind of reaction feels cold: endothermic or exothermic?
Endothermic.
Is energy being absorbed or released in this reaction:

released
These are at the beginning of a reaction and combine together.
reactants
5 observable signs of a chemical change:
new gas (fizzing, bubbling, odor), precipitate, temperature change, permanent colour change, sound or light emission
In an exothermic reaction, does it get hotter or colder?
It gets hotter
Which kind of reaction feels warm: endothermic or exothermic?
Exothermic
Is energy being absorbed or released in this reaction:
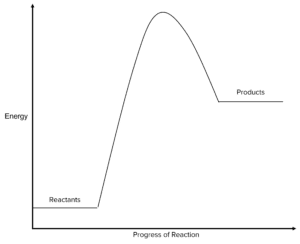
absorbed
These are created in a reaction.
products
A new chemical formula means that the properties of the new substance:
a) is different
b) is the same
different
In an endothermic reaction, does it feel warmer or cooler?
It feels cooler
Is freezing exothermic or endothermic
Exothermic
What happens to the energy of the reaction during an endothermic reaction
The reaction takes in energy from the surroundings.
Chemical reactions ALWAYS involve breaking and reforming these?
bonds
Is this a chemical change? How do you know?
Yes
Chemical formula has changed
Give one example of an exothermic reaction
Burning wood, fireworks, a campfire, etc
Is melting ice exothermic or endothermic?
Endothermic
What breaks and forms to change the energy of the reactants and products?
chemical bonds
These types of reactions will NOT occur without a source of energy.
endothermic
What does the arrow mean?
"reacts to form"
