A rectangular block of copper metal weighs 1896 g. The dimensions of the block are 8.4 cm by 5.5 cm by 4.6 cm. From this data, the density of copper is calculated to be
8.92 g/mL
A material's ability to conduct heat or electricity is classified as this type of property
Physical property
The smallest building blocks of matter
Atoms
2C8H18(l) + 25O2(g) → 18H2O(g) + 16CO2(g)
The classification for the above reaction
Combustion
These are forms of same element that contain equal numbers of protons but different numbers of neutrons in their nuclei, and hence differ in relative atomic mass.
Isotopes
These are substances that have lost electrons
Cations
An irregularly-shaped sample of aluminum (Al) is placed on a balance and found to have a mass of 43.6 g. The student decides to use the water-displacement method to find the volume. The initial volume is 25.5 mL and after the Al sample is added, the water level has risen to 41.7 mL. The student discovers that the density of aluminum is
2.69 g/mL
The ability for a metal like zinc to react with an acid like hydrochloric acid is classified as this type of property
Chemical property
These are the three subatomic particles that make up the atom
Protons, neutrons, and electrons
2 Mg (s) + O2 (g) → 2 MgO (s)
The classification for the above reaction
Synthesis or Combination
Silicon-31 contains this many neutrons
17 neutrons
This is why copper appears blue when burned and a diagram that depicts this phenomenon
Because as electrons fall back from an excited state to ground state, they emit light.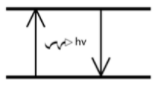
The mass of ethyl alcohol (density = 0.789 g/mL that exactly fills a 200.0 mL container is
157.8 g
Liquid water evaporating into the gas phase is an example of this type of change
Physical Change
Substances like aluminum, copper, zinc, and oxygen are often classified as
Element
PbO2 (s) → Pb (s) + O2 (g)
The classification for the above reaction
Decomposition
The element copper (Cu) has two naturally occurring isotopes, copper-63 (62.930 amu) and copper-65 (64.928 amu). The average atomic mass of copper listed on the periodic table is 63.55 amu. Of these two isotopes, this one is the most abundant for this reason
Copper-63 because the average atomic mass of the element is closer to its mass
This is the electron configuration for Arsenic
1s22s22p63s23p64s23d104p3
This volume of silver metal (density of silver = 10.5 g/mL) will weigh exactly 2500.0 g.
238.1 mL
A change to a substance that occurs by forming a new substance
Chemical Change
Substances such as carbon dioxide (CO2), water (H2O), and sodium chloride (NaCl) are classified as
Compounds
2 Al (s) + 6 HCl (aq) → 2 AlCl3 (aq) + 3 H2 (g)
The classification for the above reaction
Single Displacement
Single Replacement
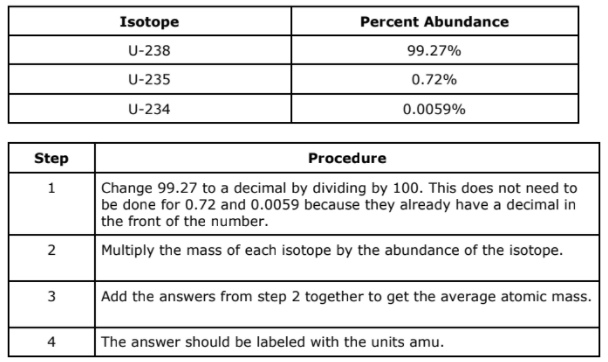 A mistake was made in this step of the process for solving for average atomic mass
A mistake was made in this step of the process for solving for average atomic mass
Step 1
This is the noble gas configuration of sulfur
[Ne]3s23p4
A student constructs composed of oil (density of oil = 0.90 g/mL), water (density of water = 1.0 g/mL), and alcohol (density of alcohol = 0.79 g/mL).The student then drops a bottle cap in the column, which comes to float in between the oil and alcohol layers. The student concludes that the density of the bottle cap is
Between 0.79 g/mL and 0.9 g/mL
This type of property does not describe the amount of matter present but describe quality likes luster and texture
Intensive property or qualitative property
Salt water is an example of this type of mixture, which has a special secondary name
Homogeneous mixture
Ca(OH)2 (s) + 2 HCl (aq) → CaCl2 (aq) + 2 H2O (l)
The classification for the above reaction
Double-Displacement
Double-Replacement
The average atomic mass for element X is 
28.08 amu
This is the electron configuration for Cl-
1s22s22p63s23p6