How many moles of O2 are contained within 12.0x1023 molecules of O2?
1.99 mol of O2 (reporting 2.0 mol of O2 is okay)
At what temperature will the solubility of KCl be 45 g/100g H2O?
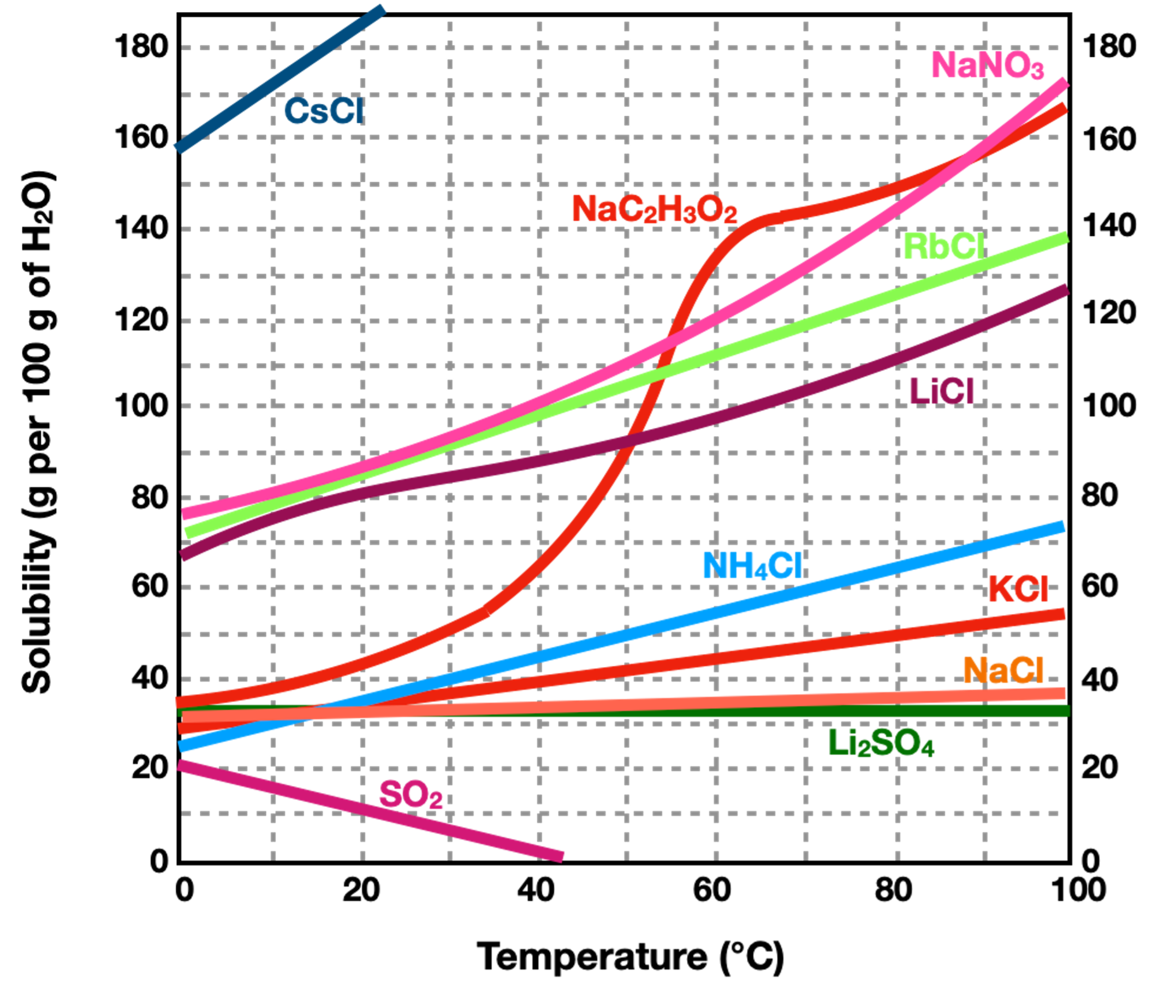
60 C
What is the electron configuration of S2-?
1s22s22p63s23p6
How many neutrons are there in cobalt-60?
Co = 60-27 = 33 neutrons
Which statement describes the transfer of heat energy that occurs when an ice cube is added to an insulated container with 100 milliliters of water at 25°C?
A. Both the ice cube and the water gain heat energy.
B. Both the ice cube and the water lose heat energy.
C. The ice cube gains heat energy, and the water loses heat energy.
D. The ice cube loses heat energy, and the water gains heat energy
C. The ice cube gains heat energy, and the water loses heat energy.
How many atoms of oxygen are contained within 4.8176x1024 molecules of O2?
Fill in the blanks:
Water, commonly known as the "universal _____________ (1)" can dissolve many substances. This is generally because water molecules are _________________ (2). However, water is not able to dissolve substances like oils, wax, lipids, and alkanes. These substances are ___________________ (3) and and therefore cannot interact effectively with water.
(1) solvent
(2) polar
(3) nonpolar
What are the names of the following compounds:
(1) Ag2SO4
(2) CBr4
(3) Fe2O3
(1) silver sulfate
(2) carbon tetrabromide
(3) iron(III) oxide
A sample of element X contains 90 percent 35X atoms, 8.0 percent 37X atoms, and 2.0 percent 38X atoms. The average atomic mass is closest to — ?
35 amu
The following text comes from a propane distributor's sales catalog.
Propane exists as a gas at standard pressure and temperatures above −44°C. However, at high pressure or lower temperatures, propane exists as a liquid.
Which of the following postulates of the kinetic molecular theory of gases does propane contradict?
A. Molecules move in straight lines until they collide with the container or other molecules.
B. Interactions among molecules, with the exception of collisions, are negligible.
C. The molecules of a gas are in constant, random motion.
D. The average kinetic energy of the gas depends only on its temperature
B. Interactions among molecules, with the exception of collisions, are negligible.
Determine the (1) empirical and (2) molecular formula for chrysotile asbestos. Chrysotile has the following percent composition: 28.03% Mg, 21.60% Si, 1.16% H, and 49.21% O. The molar mass for chrysotile is 520.8 g/mol.
(1) Mg3Si2H3O8 (empirical formula)
(2) Mg6Si4H6O16 (molecular formula)
When ethanol, C2H5OH, is added to water, it dissolves in the water by forming hydrogen bonds with the water molecules, as shown below.
Which of the following best describes ethanol?
A. A nonelectrolyte, because it is a weak base when dissolved in water
B. An electrolyte, because it forms hydrogen bonds in water that conduct electricity
C. An electrolyte, because it is highly soluble in water
D. A nonelectrolyte, because it does not dissociate into ions in water
D. A nonelectrolyte, because it does not dissociate into ions in water
--> It is organic with an OH group, but not an COOH group
Determine the chemical formulas for the following compounds:
(1) cadmium oxide
(2) xenon hexafluoride
(3) chromium(VI) phosphate
(1) CdO
(2) XeF6
(3) Cr(PO4)2
(1) Describe Rutherford's experimental design when studying atomic structure, (2) describe what conclusions Rutherford came to, and (3) why he came to those conclusions (i.e. what phenomena led him to those conclusions)
(1) He conducted the gold foil experiment (
(2) He concluded that the atom is (A) mostly empty space and that (B) there is a positively charged, densely packed nucleus
(3) He concluded this because (A) most alpha particles passed through the gold foil without interacting with it and (B) the small amount of alpha particles that did interact with the gold foil were repulsed at large angles
At which location in the Periodic Table would the most active metallic element be found?
A. In Group 1, at the top
B. In Group 17, at the top
C. In Group 17, at the bottom
D. In Group 1, at the bottom
D. In Group 1, at the bottom
A sample of 0.53 g of carbon dioxide was obtained by heating 1.31 g of calcium carbonate. What is the percent yield for this reaction?
91.9%
A 1.50 M sugar solution is prepared by mixing sucrose C12H22O11 with enough water to create a 0.850 L solution. The molar mass of sucrose is 342.296 g/mol.
What mass of sucrose must be added to make this solution?
436 g
Describe what the electrons are doing in the following bond types: (1) metallic, (2) ionic, (3) nonpolar covalent, (4) polar covalent
(1) delocalized as a "sea of electrons"
(2) transferred from cation to anion
(3) equally shared
(4) unequally shared
To determine if a salt is soluble or insoluble, chemists often memorize solubility rules. They may also consult a solubility chart, which shows the solubility of different combinations of cations and anions.
Which of the following salts are insoluble in water at 25°C?
Choose 2 answers:
- A) calcium fluoride
- B) zinc sulfate
- C) lead(II) acetate
- D) aluminum phosphate
- E) lithium carbonate
A) calcium fluoride
D) aluminum phosphate
As the elements of Group 16 of the Periodic Table are considered in order from top to bottom, the covalent radius of each successive element increases. This increase is primarily due to an increase in —
A. the number of protons occupying the nucleus.
B. atomic number.
C. mass number.
D. the number of occupied electron shells
D. the number of occupied electron shells
(1) Calculate the mass of magnesium oxide possible if 2.40 g Mg reacts with 10.0 g of O2. (2) What is the limiting reagent?
(1) 1.58 g of O2
(2) Mg
While experimenting with double replacement reactions, a lab group adds a solution of barium nitrate to a solution of ammonium sulfate.
(1) Write the full, balanced chemical equation with the phases.
(2) Will this reaction occur?
(1) Ba(NO3)2 (aq) + (NH4)SO4 (aq) --> 2 NH4NO3 (aq) + BaSO4 (s)
(2) Yes
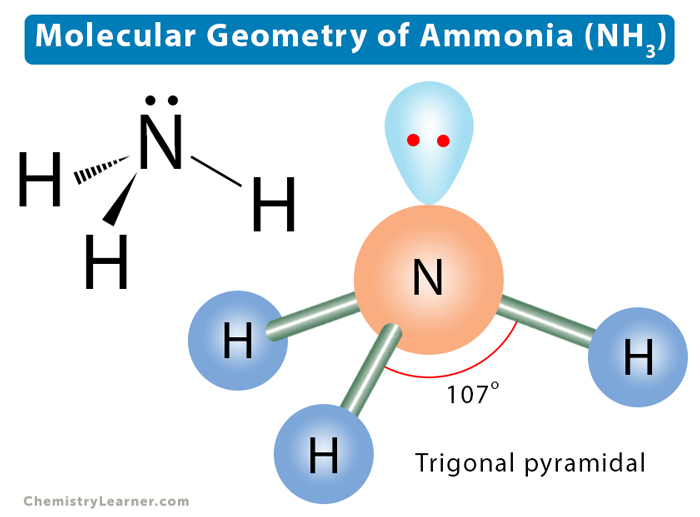
(1)Report the total number of valence electrons in NH3, (2) draw the line-bond Lewis structure for NH3 with the correct molecular geometry, and (3) report the name of the molecular geometry it exhibits.
NH3 has 8 total valence electrons and forms a trigonal pyramidal electron geometry.
An atom emits a photon with energy of 7.13 electronvolts (eV). What is the wavelength of the photon, in nm?
174 nm
The reaction of photosynthesis is shown.
6 CO2 + 6 H2O + heat --> C6H12O6 + 6 O2
Which graph shows the change in energy of this reaction?
A.
B.
C.
D.
D.