What are the 3 things needed when drawing a Bohr-Rutherford diagram?
Protons, neutrons & electrons
Explain the difference between a qualitative property and a quantitative property.
A qualitative property cannot be measured but can be identified by colour, smell or texture.
A quantitative property can be measured or counted, like mass, length & temperature
What is the scientific name of a mechanical mixture?
Hetergeneous mixture
What is density?
A measure of how many particles are packed into a given space.
How to figure out the number of neutrons in an element
Subtract the atomic number from the atomic mass
Draw Lithium as a Bohr-Rutherford Diagram
2 on inner shell
1 on valence shell
Classify whether this is a physical or chemical change:
Sugar, eggs, and flour are mixed and baked into cookies
Chemical change.
Hydrogen is an example of a...
Element
How is density calculated?
Dividing an object's mass by its volume
How many elements total in the periodic table?
118
Draw Neon as a Bohr-Rutherford diagram
2 on inner shell
8 on valence shell
List 2 physical properties of a candle
Colour-
Texture-
Shape-
Melting point-
Etc
CO2 is an example of a...
Compound
Calculate the density of sulfuric acid if 35.7mL of the acid has a mass of 65.17g
The density of the sulfuric acid is 1.83g/mL
What are the two main categories that separate elements of the periodic table?
Metals & Non-metals
Draw Chlorine as a Bohr-Rutherford Diagram
2 on inner shell
8 on middle shell
7 on valence shell
Classify whether this is a physical or chemical change:
Orange drink crystals are stirred into a pitcher of water
Physical change.
What are the 3 states of matter?
Solid, Liquid & Gas
What method is used to calculate the density of an object?
BONUS: What does each letter of the acronym stand for?
GUESA
G: given
U: unknown
E: equation
S: subsitute & solve
A: answer
How many atoms of each element are in this formula?
NaHCO3
Sodium: 1 atom
Hydrogen: 1 atom
Carbon: 1 atom
Oxygen: 3 atom
Draw Argon as a Bohr-Rutherford Diagram
2 on inner shell
8 on middle shell
8 on valence shell
Desrcibe several physical and chemical properties of Carbon Dioxide.
Physical: colourless, odorless gas at room temp, denser than air, nonflammable
Chemical: does not burn, does not support combustion, slightly acidic, reacts with water and bases
List the 4 main ideas of the particle Theory
1. All matter is made up of tiny particles
2. All particles of one substance are the same.
3. Particles are in constant motion.
4. There are attractive forces between particles.
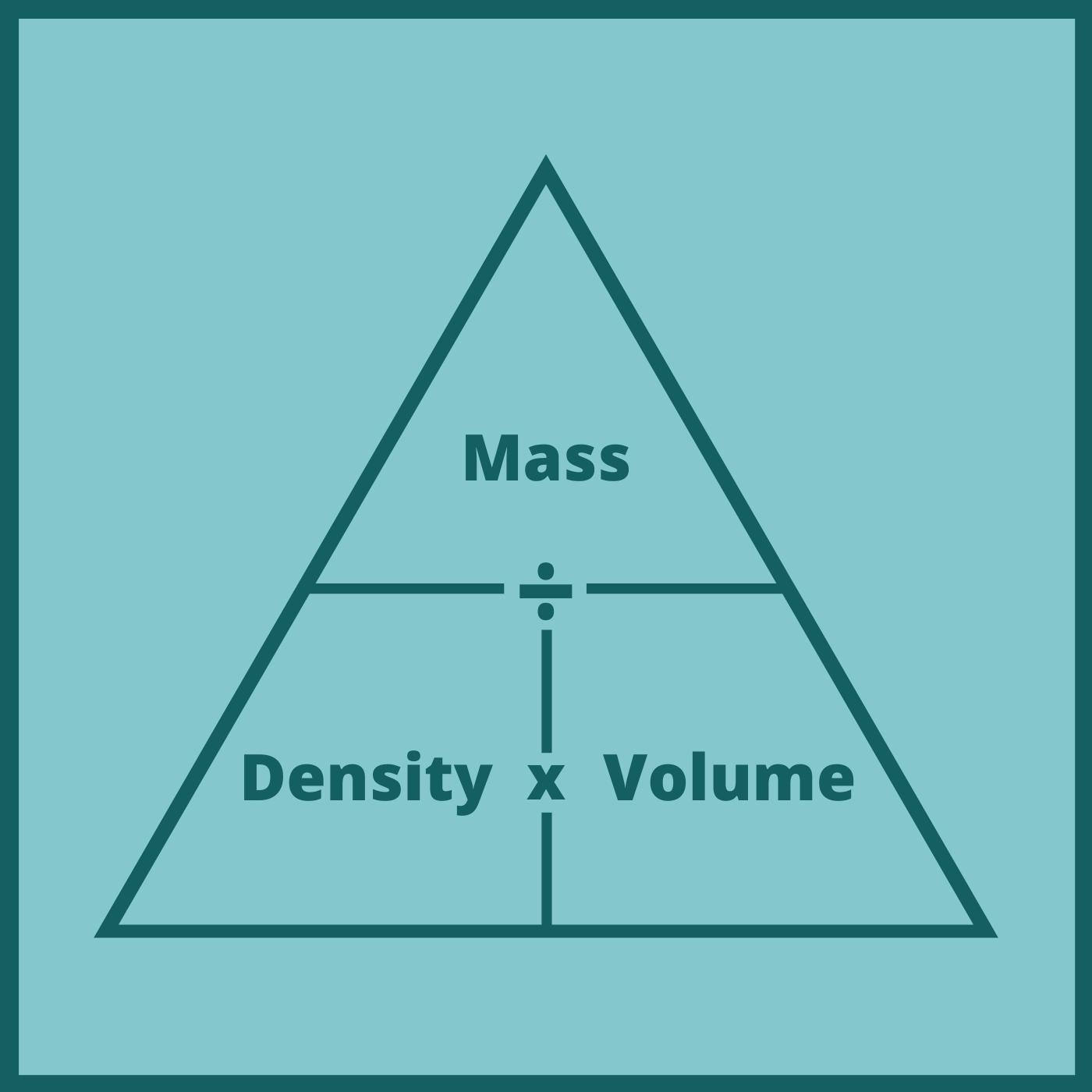
Use the magic triangle formula to show the formulas for density, mass & volume
What group are Halogens in & what group are Noble Gases in?
Halogens: group 17
Noble Gases: group 18