Stoichiometry
Which of these are NOT a unit of pressure?
a. pounds per square inch
b. kilopascals
c. kilowatts
d. atmospheres
e. mm Hg
c, kilowatts
True or false: gas particles sometimes stop moving to take a break.
False
Kelvin, K
What do the 1 and 2 mean in the combined gas law equation?
1 = before or initial
2 = after or final
50 points for each answer.
A mixture of H2 at 2.33 atm and N2 at 0.77 atm is in a container. What is the total pressure in the container?
Ptotal = 3.10 atm
Mg (s) + 2 HCl (aq) --> MgCl2 (aq) + H2 (g)
How many moles of gas product can be made from 3.45 moles of HCl?
1.73 moles
Ms. Binkley's car ties should be at 36.0 psi. Convert to atm.
2.45 atm
Gas particles can compress because _________.
There is a lot of space between particles.
Want to find P in torr given V = 0.75 L, n = 0.345 moles, and T = 435.3 oC. What value of R would you use?
One of two answers is ok:
Option 1: 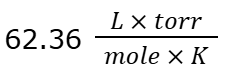
Option 2: change P to atm, then use

P1 = 8.94 atm
V1 = 17.2 L
V2 = 2.71 L
Find P2. Report answer with the correct units.
P2 = P1V1/V2
P2 = 56.7 atm
In a gas mixture of He and Ne, the total pressure is 335 torr and the partial pressure of He is 0.228 atm. What is the partial pressure of Ne?
PNe = 162 torr
Cl2 (g) + PCl3 (g) --> PCl5 (g)
Total pressure is 4.63 atm. PPCl3 is 2.5 atm. Total amount of reactant moles is 2.35.
a. Find the moles of PCl3.
b. How many moles of PCl5 can be made from using up all the PCl3?
a. (100 points) = 1.27 moles PCl3
b. (100 points) = 1.27 moles PCl5
Convert 778.0 torr to kPa.
103.7 kPa
True or false: real gases do not have intermolecular force attractions.
False. Ideal gases do not, but real gases DO have intermolecular force attractions.
Given:
P = 98,100 Pa
n = 0.839 moles
T = 425.0 oC
Find V, in cubic meters (m3).
V = 0.0496 m3
Initially have 0.613 moles at 6.1 L. Then add more gas, ending up with 0.931 moles. What is the final volume? Report answer with the correct units.
V2 = V1n2/n1
V2 = 9.26 L
The partial pressure of F2 in a mixture of gases is 302 torr. The total pressure is 1.00 atm. What is the mole fraction of F2?
0.397
H2 (g) + I2 (g) --> 2 HI (g)
Total pressure is 4.28 atm, total reactant moles is 5.00. PI2 is 1.84 atm.
a. Determine the moles of I2.
b. How many moles of HI can be made when all the I2 is used up?
a. (150 points) 2.15 moles I2
b. (150 points) 4.30 moles HI
When we watched the video about the free diver, why did her lungs compress as she dove deeper into the ocean?
P = (density) * gravity * height
As she goes down, more height of water above her, so more pressure.
*they need something along those lines for the credit*
Name one reason from the kinetic molecular theory that thinking of gas particles like billiard balls is helpful.
Need to mention one of these:
a.) Gas particles move in straight lines, until they collide with something. Then they bounce off at an angle.
or
b.) When gas particles collide, they do not compress or deform or lose energy because of the collision. This is called an elastic collision.
Given:
P = 6.46 atm
V = 3.80 L
n = 0.875 moles
Find T in Kelvin, then change to Celsius.
200 points: T = 342 K
200 more points: T = 68.7 oC
P1 = 2.45 atm
P2 = 7.67 atm
T1 = 126.5 oC
Find T2, in oC.
Remember, still need T in Kelvin for the combined gas law equation.
T2 = T1P2/P1
T2 = 1251 K (200 points) = 978 oC (200 more points)
If 60.0 L of nitrogen is collected over water at 40.0 °C when the atmospheric pressure is 760.0 mm Hg, what is the partial pressure of the nitrogen? At 40.0 °C, the vapor pressure of water is 7.3814 kPa.
PN2 = 704.6 mmHg
Zn (s) + 2 HNO3 (aq) --> Zn(NO3)2 (aq) + H2 (g)
Want to make 1.25 L of H2 gas at 23.0 oC and 1.15 atm.
a. How many moles of H2 are to be made?
b. How many moles of HNO3 are needed to make the wanted amount of H2?
a. (200 points) 0.0591 moles H2
b. (200 points) 0.118 moles HNO3
Which would you expect to have a smaller pressure and why? Explain in terms of what is happening to the particles.
Option A: a 10.0 L balloon filled with 6.75 grams of helium, at 23.0 degrees C.
Option B: a 10.0 L balloon filled with 6.75 grams of helium, at 45.0 degrees C.
Option B because the helium atoms would move faster, and have more collisions with the wall.
*They need to mention collisions with the wall and the effect of the temperature on the atoms for the credit. If they solve an equation, they did not explain in terms of the particles.
For two of these relationships, explain if it's direct or indirect. Explain means more than say direct or indirect, need to talk about on the particle level what's happening to make that happen.
a. moles and volume
b. temperature and pressure
c. volume and pressure
d. moles and pressure
a. moles and volume: direct. More particles take up more space.
b. temperature and pressure: direct. Faster moving particles have more collisions with the wall than slower moving particles.
c. volume and pressure: indirect. As the volume increases, there are less collisions with the wall. (Or the opposite).
d. moles and pressure: direct. More particles have more collisions.
For the points: need to mention direct or indirect, and a reason. The reason should be along the same lines as what I've said here.
Given:
P = 8.37 atm
V = 14.9 L
T = 545.1 oC
The gas is argon, Ar.
Find the moles of gas. Then change to grams.
250 points: n = 1.86 moles Ar
250 more points: 74.2 grams Ar
500.0 liters of a gas in a flexible-walled container are prepared at 0.921 atm and 200.0 °C. The gas is placed into a tank under high pressure. When the tank cools to 20.0 °C, the pressure of the gas is 30.0 atm.
What is the final volume of the gas?
V2 = T2P1V1/P2T1
T in Kelvin, btw
V2 = 9.51 L
A mixture of 2.20 moles of H2, 3.70 moles of NH3, 4.50 moles of CO2, and 5.10 moles of N2 exert a total pressure of 800.0 torr. What is the partial pressure of each gas?
125 points per gas that's correct.
H2 = 114 torr
NH3 = 191 torr
CO2 = 232 torr
N2 = 263 torr
2 Na (s) + Cl2 (g) --> 2 NaCl (s)
Have 5.50 L of Cl2 at 1.25 atm and 22.0 oC.
a. How many moles of Cl2 is there?
b. How many moles of NaCl can be made?
c. Convert the moles NaCl to grams.
a. (150 points) 0.284 moles Cl2
b. (150 points) 0.568 moles NaCl
c. (200 points) 33.2 g NaCl