What is happening at the triple point of a substance?
All three phases (solid, liquid, gas) exist at equilibrium
What happens to pressure if force increases but area stays the same?
pressure increases
What happens to a gas's volume if the temperature increases at constant pressure? (increase, decrease, stay the same)
volume increases
How does increasing temperature affect gas density at constant pressure?
density decreases
What is Dalton's Law
Pa = Xa*Ptot
What 2 phases become indistinguishable at the critical point?
gas and liquid
Why do snowshoes reduce pressure on the snow?
they increase area and thus reduce pressure
A gas at constant temperature doubles in volume (L). What happens to pressure?
it halves
Why are heavier gases more dense than lighter gases?
Higher molar mass
A mixture has gases at 200. torr and 300. torr. What is total pressure in atm?
0.658 atm
what phase is water in at 150 oC and 0.5 atm?
gas
A force of 100 N acts on an area of 2.0 m². What is the pressure in KPa?
0.05 KPa
Calculate pressure: n = 1.50 mol, V = 10.0 L, T = 300 K
3.69 atm
A gas has mass 10.0 g and volume 5.0 L. What is density?
2.0 g/L
A gas has hydrogen makes up 16% of a mixture at 4.62 atm. What is its partial pressure?
0.74 atms
What phase change occurs at -50 oC when water goes from 0.06 atms to 0.00006 atms?
sublimation
A gas exerts 1.20 × 10⁵ Pa on a surface that is 0.67 m by 0.75m. What force is applied?
6.0 x 104 N
A gas: P₁ = 1.00 atm, V₁ = 2.00 L, T₁ = 300 K → V₂ = 4.00 L, T₂ = 350 K. Find P₂
0.58 atm
A gas has density 1.20 g/L at 1.00 atm and 300 K. What is density at 2.00 atm and same temp?
2.40 g/L
In a 5682 mL container, there is 56.2 g of CO2, 44.5g N2, and 17.0 g H2 at 452 K. What is the total pressure in the container
73.7 atms
What is the final phase of matter: start: -75 oC, 17 atm 2)temp rises to -40 oC 3) pressure rises to 80 atms, 4) temp rises to 40 oC 5) pressure plummets to 1 atm
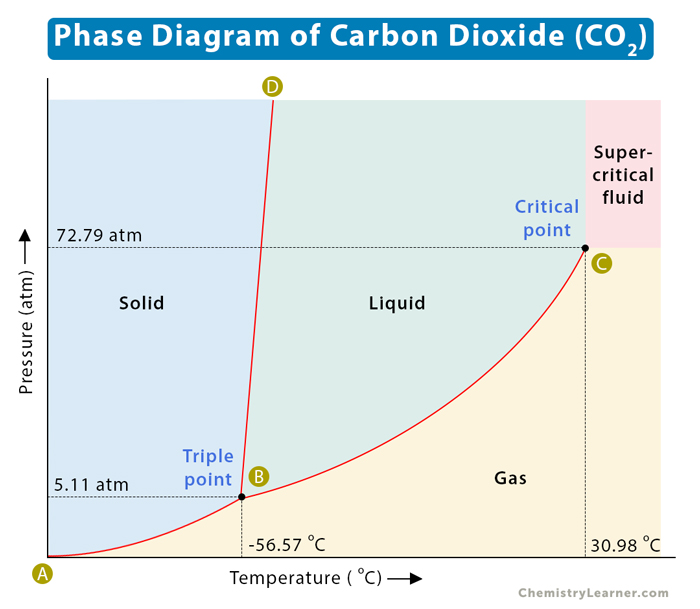
gas
A circular surface has radius 0.100 m and pressure of 2.00 × 10⁵ Pa. What force is exerted?
6.28 × 10³ N
A gas originally existed in a 1652 mL tank at STP. It is pumped into a much smaller tank (534 mL) where the new temperature was found to be 451 oC. What is the new pressure?
8.20 atms
A gas has density 1.00 g/L at 300 K and 1.00 atm. What is density at 600 K and 0.50 atm?
0.25 g/L
Propane combusts according to the balanced reaction:
C₃H₈(g) + 5 O₂(g) → 3 CO₂(g) + 4 H₂O(g)
A sealed 12.0 L container at 350 K contains 1.20 mol C₃H₈ and excess O₂. After the reaction goes to completion, the water vapor is condensed and removed, leaving only CO₂ and excess O₂ in the container. What is the partial pressure of CO₂ in the container?
8.62 atm
