Pressure
The force per unit area
on a surface.
Absolute Zero
The temperature –273.15°C
Is given a value of zero in the Kelvin temperature scale. ( 0 K )
What is the equation of Absolute zero?
K=273.15+°C
Gay-Lussac’s law of combining volumes of gases
States that at constant temperature and pressure, the volumes of gaseous reactants and products can be expressed as ratios of small whole numbers.
What is the equation of Ideal gas law?
PV = nRT
Newton
The force that will increase the speed of a one-kilogram mass by one meter per second each second that the force is applied.
Boyle’s law
States that the volume of a fixed mass of gas varies inversely with the pressure at constant temperature.
What is the equation of Gay-Lussac's law?
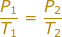
Avogadro’s law
The law states that equal volumes of gases at
the same temperature and pressure contain equal numbers of molecules.
What is the equation of Avogadro's law?
V=kn
Barometer
•A device used to measure atmospheric pressure.
Charles’s law
States that the volume of a fixed mass of gas at constant pressure varies directly with the Kelvin temperature.
What is the equation of Boyle's law?
P1 V1 = P2 V2
The ideal gas law
The mathematical relationship among pressure, volume, temperature, and number of moles of a gas.
What is the equation of Avogadro's constant?
(6.022× 1023)
Dalton’s law of partial pressures
States that the total pressure of a gas mixture is the sum of the partial pressures of the component gases.
Gay-Lussac’s law
States that the pressure of a fixed mass of gas at constant volume varies directly with the Kelvin temperature.
What is the equation of Charles's law?
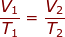
The standard molar volume of a gas
The volume occupied by one mole of gas at STP.
What is the equation of Ideal gas law constant?
R=PV/nTn
Vapor Pressure
The pressure of a vapor in contact with its liquid or solid form.
Combined gas law
Expresses the relationship between pressure, volume, and temperature of a fixed amount of gas.
What is the equation of the Combined gas law?
P1V1 / T1 = P2V2 / T2
Ideal gas constant
In the equation representing the ideal gas law.
What is the equation of Graham's law of effusion?