How does the energy of an orbital change as the distance from the nucleus increases
energy increases
What row are Halogens in
Row 17 or 2nd row to the right
What is a covalent bond
Sharing of electrons between 2 elements with similar electronegativity
What is a decomposition reaction
Single reactant breaks down
2HgO(s) → 2Hg(l) + O2(g
Chem Fun Fact
The only solid elements that assume liquid form at room temperature are bromine and mercury.
What is the Heisenberg uncertainty principle
It is impossible to know the momentum and position simultaneously.
What column are alkaline Earth Metals in
What is an ionic bond
transfer of one or more electrons from an element with a relatively low ionization energy to an element with a relatively high electron affinity
Molarity equation
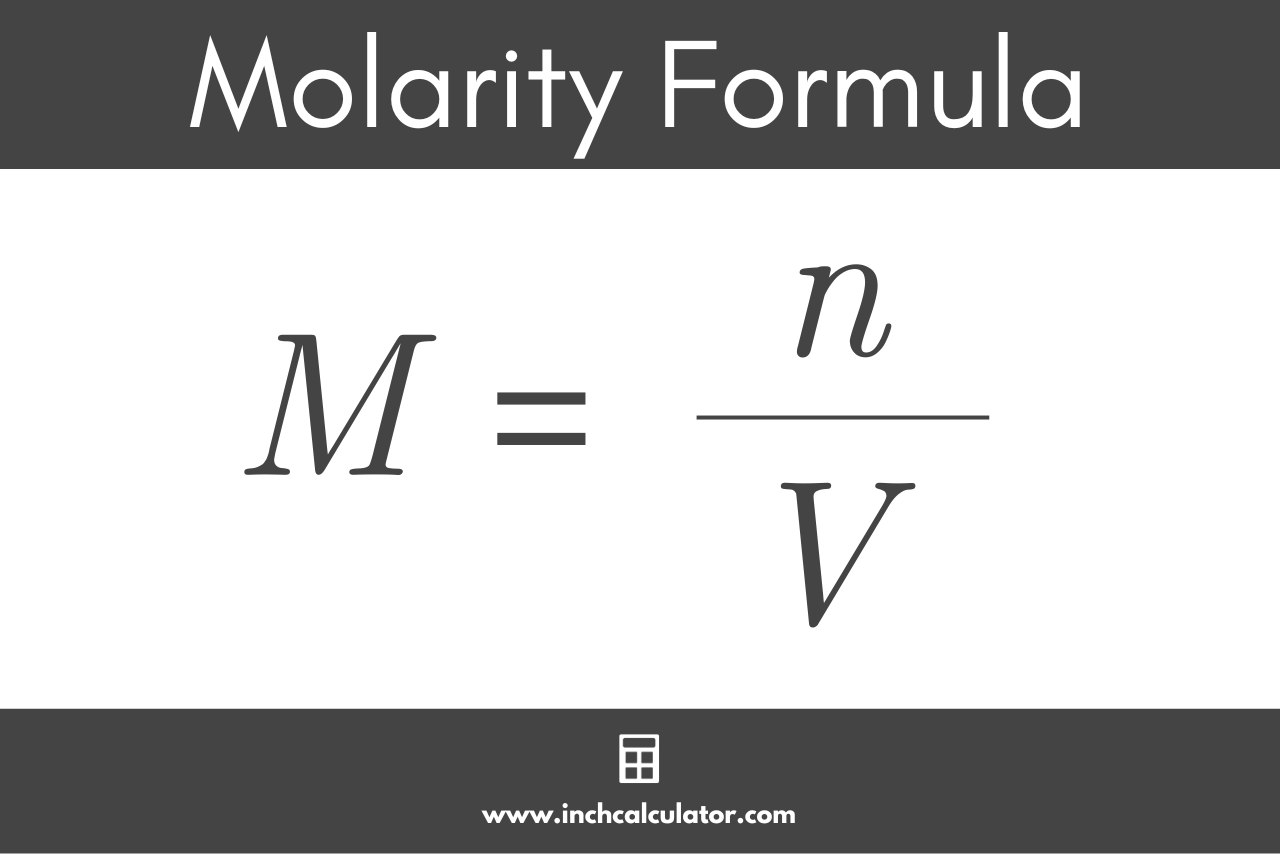
Ava and Brynn fun fact
The first photo I have in my phone of you was from September 3rd 2022
that's 1,297 days
That's 42.6 months ago
or 3.55 years ago
What is the equation for light energy
E = hc/ wavelength
What element is the most electronegative
F
Rank the strength of the IMF: Hydrogen, LDF, and Dipole-Dipole
Strongest: Hydrogen
Dipole Dipole
Weakest: LDF
What is a combustion reaction
usually a hydrocarbon, and O2(g) Commonly forms CO2 and H2O
CH4 (g) + 2O2 (g) → CO2 (g) + H2 O (g)
regular fun fact
Contrary to popular belief, there aren’t “57 varieties” of Heinz ketchup (and never were).
Company founder H.J. Heinz thought his product should have a number, and he liked 57.
What does diamagnetic mean in orbitals
All electrons are paired REPELLED by an external magnetic field
What direction on the periodic table does atomic size increase
Left and down
What is the charge of a cation
positive
What are the 2 products of a neutralization reaction
a salt and H2O
Chem joke
What is Cole’s Law?
Thinly sliced cabbage.
What is the atomic orbital of C
1s2 2s2 2p2
What direction on the periodic table does ionization increase
Right and up
What is the most likley shape of an sp3 hybridized molecule
Tetrahedral or Trig Pyramidal
What is the formula for Dihydrogen Phosphate
H2PO4
Cute animal picture
