Acid rain is an example of which type of weathering?
A. Chemical
B. Physical
A. Chemical
Which subatomic particle has a positive charge?
A. Proton
B. Neutron
C. Electron
A. Proton
Which of the following is an example of water pollution?
A. A pond with an overpopulation of fish
B. A lake that is littered with plastic bottles
C. A saltwater bay
B. A lake that is littered with plastic bottles
True or false:
Atoms can combine to form new substances
True
Why is water considered the "universal solvent"?
It dissolves more substances than any other liquid
What is the center of an atom called?
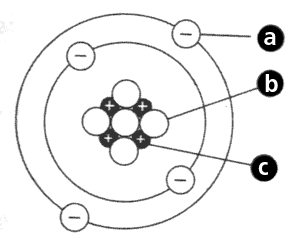
The Nucleus
Which of the following actions would be most safe and effective for conserving water?
A. Showering less often
B. Drinking mostly juice instead of water
C. Using a reusable water bottle instead of a plastic bottle
D. Only washing the dishes three times per week
C. Using a reusable water bottle instead of a plastic bottle
When different types of atoms combine, they form a(n)
A. Element
B. Compound
B. Compound
Water flows into cracks in a rocky surface and then freezes. What would likely happen to the rock as as a result?
The rock would break into smaller pieces because water expands when it freezes
What two subatomic particles are located in the nucleus of an atom?

Protons and Neutrons
Which two uses of water most directly affect public health and safety?
A. Filling up swimming pools
B. Washing hands
C. Generating electricty for hydropower plants
D. Watering house plants
E. Mopping the floors
F. Washing cars
B. Washing hands and C. Generating electricty for hydropower plants
What is the chemical equation for H2O?
H2 + O = H2O
What property of water allows water striders to stay on the surface of water?
Surface Tension
Which subatomic particle orbits around the nucleus?

Electrons
What percent of freshwater is available for humans to use at any time?
1%
O2 and H4 are examples of _____
A. Compounds
B. Elements
B. Elements
Which property of water allows ice to float in water?
Density
What kind of charge does each subatomic particle have?
Proton
Neutron
Electron
Proton - Positive
Neutron - Neutral
Electron - Negative
Where is most of earth's freshwater stored?
In ice caps/glaciers
______ are made up of different types of atoms
A. Compounds
B. Elements
A. Compounds