B, Si, Ge, As, Sb, and Te.
What are the metalloids?
The trend for increasing atomic radius on the periodic table.
What is down and to the left?
The full electron configuration for silicon (Si).
What is 1s22s22p63s23p2?
A description of the relationship between wavelength and frequency of electromagnetic radiation.
What is inverse?
(As one increases, the other decreases.)
(c =λν)
A property of materials to be weakly affected by magnetic fields due to unpaired electrons resulting in a nonzero spin.
What is paramagnetism?
The category of elements that can have various oxidation states (charges).
What are the transition metals?
The most electronegative element on the periodic table.
What is fluorine?
The noble gas configuration for silver (Ag).
What is [Kr] 5s14d10?
The relationship between the energy of a photon and its frequency.
What is E = hν?
The orbital diagram and type of magnetism of magnesium (Mg).
What is 1s22s22p63s2 and diamagnetic?
The "group" of elements known for being highly radioactive.
What are the actinides?
The most electronegative of the elements Cl, S, Br and N.
What is Cl?
The shell and subshell of an electron with the quantum numbers {5,3,-2,½}.
What is 5f?
The wavelength of a photon (in nanometers) with a frequency of 5.45 × 1014 Hz.
What is 550 nm?
The type of magnetism experienced by group 12 elements.
What is diamagnetism?
The predominant property of the group of elements containing Rn.
What is (inert/full valence shell)?
The order of the elements Kr, Ca, Ar, S, B by increasing atomic radius.
What is B < Ar < S < Kr < Ca?
The noble gas configuration for bismuth (Bi).
What is [Xe] 6s24f145d106p3?
The energy of a photon with a frequency of 2.91 × 1014 Hz.
What is 1.93 × 10-21 J?
What is (Co, Fe, Ni, Gd, Tb, Dy)?
The smallest element of a group of elements known for their ability to form basic (alkaline) solutions in water?
What is beryllium (Be)?
The order of the elements O, Br, Ne, He, F by increasing electronegativity.
What is He = Ne < Br < O < F?
A set of quantum numbers that could describe the last electron added to chlorine.
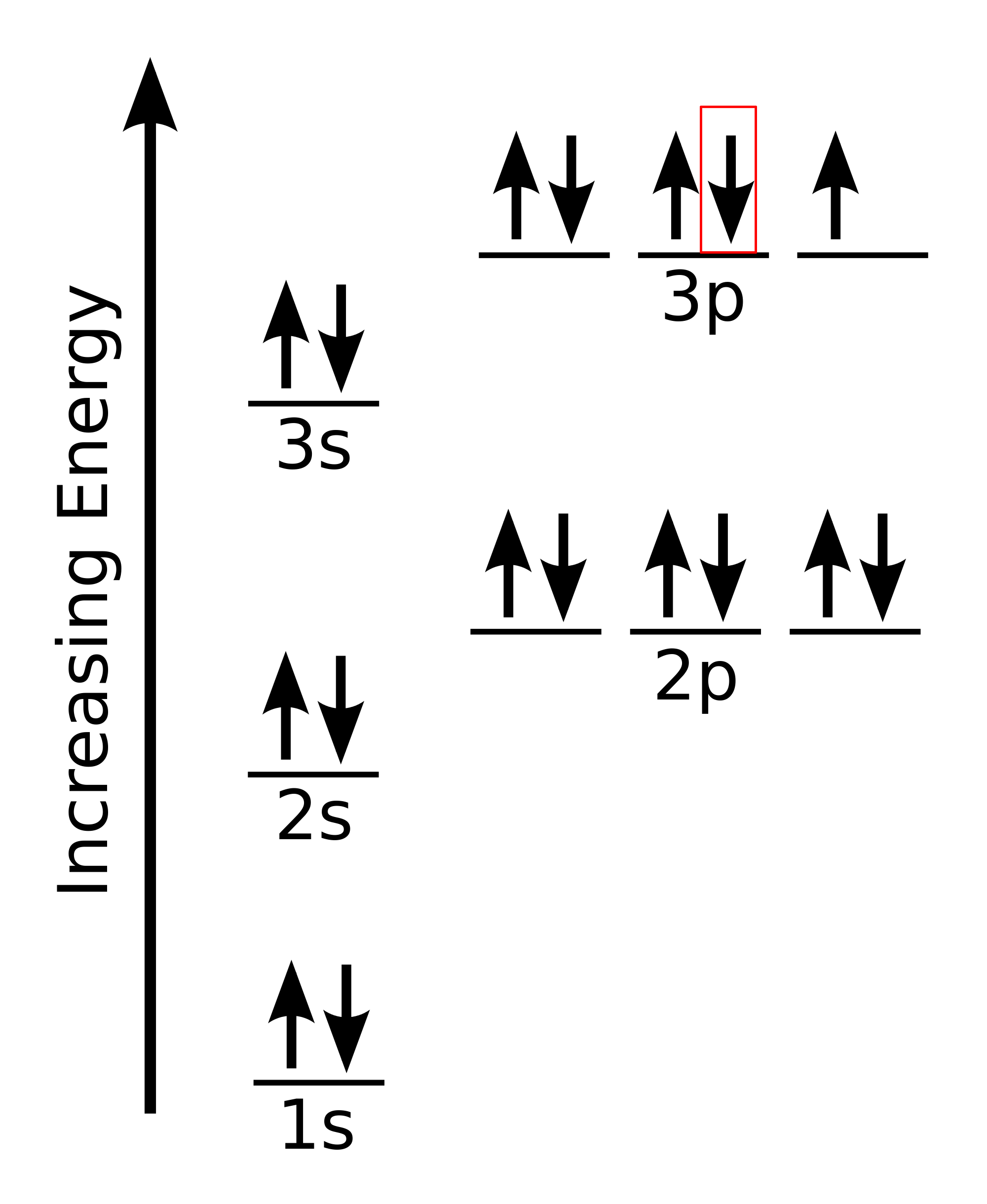
What is {3,1,0,-½}?
(possible values of mℓ = -1, 0, 1)
(possible values of ms = ½, -½)
The wavelength of a photon in nanometers needed to excite a hydrogen atom from n=2 to n=5, requiring an energy of 1.550 × 10-19 J.
What is 1281 nm?
Two diamagnetic elements with the outermost energy level n=2.
What are beryllium (Be) and neon (Ne)?