Two isotopes of an element must differ in number of _____________.
a. protons
b. neutrons
c. electrons
d. shells
B
What is Avogadro's number?
6.02 x 1023
What is the name of the leading number in front of a chemical compound in a reaction (ex: the 2s in 2H2 + O2 --> 2H2O)?
coefficient
What are the two parts of every solution?
solute and solvent
What is the relationship between the [H+] and [OH-] in a sample of pure water?
equal
How many neutrons does the following isotope of iodine contain?
I-125
72
What is the gram formula mass (molar mass) of C2H4 rounded to the hundredths place?
28.06 g/mol
What is the mole ratio between N2 and NH3 in the Haber reaction below?
N2 + 3H2 --> 2NH3
1:2
What is the maximum mass of sucrose that can be dissolved in 100 mL of water at 60oC?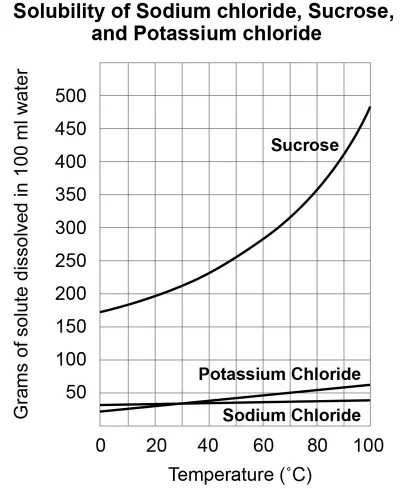
~280 g
Which of the following is an arrhenius acid?
a. NaOH
b. NaCl
c. C2H4
d. HF
D
Gallium has only two stable isotopes: Ga-69 and Ga-71. The average atomic mass of gallium is 69.72 amu. Gallium-based LEDs contain a mixture of these two naturally occurring isotopes. Based on its average atomic mass, which isotope is more prevalent in a gallium LED?
a. Ga-69
b. Ga-71
c. both contribute equally
d. impossible to know with the given information
A
Convert 12 g of water to moles of water. Round to the hundredths place.
0.67 moles
How many moles of ammonia (NH3) are produced when 4.2 moles of H2 react with excess N2?
N2 + 3H2 --> 2NH3
2.8 moles of NH3
What is the molarity of an NaCl (aq) solution when 58.44 g of NaCl is dissolved in a 0.5 L solution?
2 M
Finish the neutralization reaction and balance.
H2SO4 + KOH -->
H2SO4 + 2KOH --> 2H2O + K2SO4
List the # of protons, neutrons, and electrons in a chloride ion (Cl-) with a mass number of 36
protons -17
neutrons - 19
electrons -18
Convert 3.40 moles of NaCl to grams of NaCl. Round to the nearest whole number.
199 g
2 NaCl → 2 Na + Cl2
How many grams of sodium chloride (NaCl) are required to make 0.32 moles of chlorine (Cl2)?
37 g NaCl
What is the concentration in parts per million of an AgCl(aq) solution that contains 1.2 x 10-3 grams of AgCl in 800. grams of the solution?
1.5 ppm
What is the [H+] of a substance with a known [OH-] of 3.4 x 10-10?
2.9 x 10-5 M
Calculate average atomic mass and identify the element using a periodic table based on the following data:
Isotope 1: 68.926 amu, 60.11% abundance
68.926 amu, 60.11% abundance
Isotope 2: 70.925 amu, 39.89% abundance


69.72 amu, Gallium (Ga)
How many molecules of water are in 230 g of water?
7.7 x 1024 molecules
2 Ga2O3 → 4 Ga + 3 O2
How many grams of oxygen (O2) can be produced from 15.2 grams of gallium oxide (Ga2O3)?
3.89 g
If 25.0 mL of a 2.19 M solution are diluted to 72.8 mL, what is the final concentration?
0.752 M
Calculate the [OH-] of a solution with a known pH value of 5.6.
4.0 x 10-9 M