What is heat?
Mode of motion, transfer of energy from warmer objects to colder objects
The heat lamp is an example of what mode of heat transfer?
radiation
What happens to particles when materials warm up?
The particles move faster and spread further apart.
What is this state?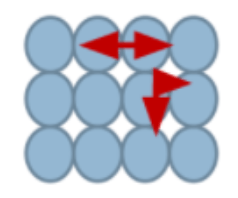
solid
What is this state?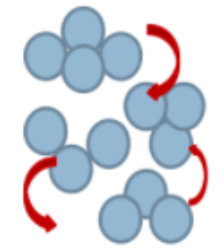
Liquid
What is kinetic energy?
Energy that all particles have. Energy of the movement of the particles.
In what states of matter would we find convection currents?
liquids and gases (fluids)
The force of attraction is greatest between particles of a...
solid
What is this state? 
Gas
What is the particle arrangement of a liquid?
Small groups of particles clumped together. Some space between groups of particles.
What is temperature?
The measure of the average kinetic energy of the particles of a substance.
Conduction
Which of physical states of water contain particles with the highest amount of movement?
Water vapour
When scientific term is used when a gas turns back into a liquid? 
Condensation
What scientific term do we use when a liquid turns into a gas?

Evaporation
What is thermal energy?
The total kinetic energy and energy of attraction of all the particles in the material.
Steam coming off of a cup of tea is what mode of heat transfer?
Convection
You pound a nail and find that it is hot to the touch afterwards. What is happening to the particles?
The particles in the nail have more kinetic energy after pounding.
The orange juice turning into popsicles would be freezing. The popsicle turning back into orange juice would be melting.
What is the particle attraction of a gas, liquid and solid?
Solid has strong particle attraction.
Liquid has medium particle attraction.
Gas has weak particle attraction.
What is thermal expansion and thermal contraction?
Thermal expansion is an increase in the volume of a substance caused by heating. Thermal contraction is a decrease in the volume of a substance caused by cooling.
Define conduction, convection, and radiation.
Conduction - Heat is moved through a warmer object that is touching a colder object.
Convection - Heat is circulated through fluids such as air (gas) or water (liquid).
Radiation - Heat is moved or transferred between two objects that are not touching. Energy from the source moves outward in a wave.
What happens when a faster particle bumps into a slower particle?
The faster particle will transfer its energy to the colder particle and slow down. The slower particle will warm up from the heat of the faster particle and speed up. 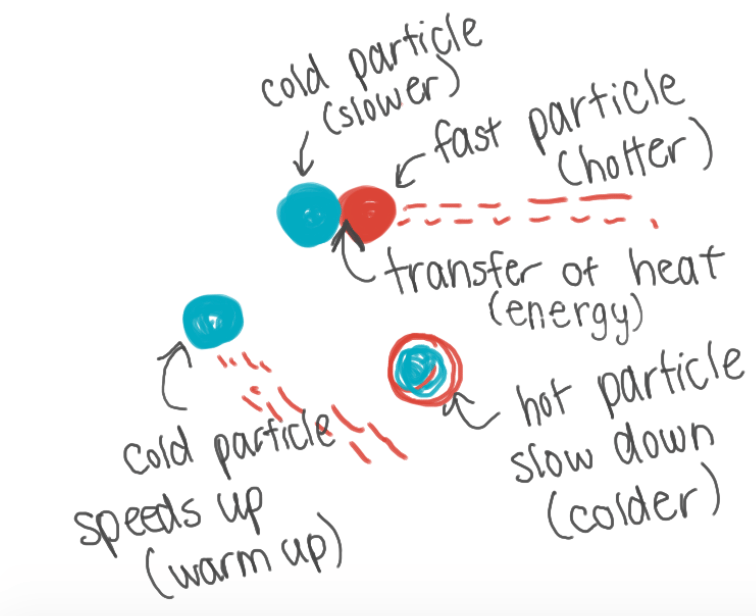
What is the particle motion of a solid compared to a gas?
The particles of a solid vibrate and do not move around. The particles of a gas freely move and roll past each other.
What is the kinetic energy of a gas, liquid and solid?
Gas has high kinetic energy.
Liquid has medium kinetic energy.
Solid has low kinetic energy.