What type of bond involves the transfer of electrons?
ionic
What type of reaction is: A + B → AB?
synthesis
Name: CO₂
carbon dioxide
What do Lewis dot structures show?
Valence electrons
What law requires equations to be balanced?
law of conservation of matter/mass
What is the mole used for?
counting extremely large amounts (usually atoms/particles/molecules)
What is percent yield?
actual amount of substance produced compared to theoretical amount
What type of bond forms between two nonmetals?
covalent
What type of reaction is: AB → A + B?
decomposition
Name: NaCl
sodium chloride
How many valence electrons does oxygen have?
6
Balance: H₂ + O₂ → H₂O
2H₂ + O₂ → 2H₂O
How many moles are in 18.0 g of H₂O?
1 mol
Write the percent yield formula

Why do atoms form bonds? (Be specific about energy/stability)
to fill their valence shells/to fulfill the octet rule/to become stable
Identify the reaction type:
Zn + CuCl₂ → ZnCl₂ + Cu
single replacement
Write the formula: dinitrogen tetroxide
N2O4
Draw the Lewis structure for H₂O

Balance: Fe + O₂ → Fe₂O₃
4Fe + 3O₂ → 2Fe₂O₃
What is the mole ratio in:
2H₂ + O₂ → 2H₂O
2 mol of H2 : 1 mol of O2 : 2 mol of H2O
Calculate percent yield: actual = 40g, theoretical =50g
80%
Explain the difference between ionic and covalent bonding in terms of electrons/electronegativity
ionic - electrons transferred bc large differences in electronegativity (one atom pulls all valence electrons very close to its nucleus)
Covalent - electrons shared bc smaller differences in electronegativity (one atom may pull all valence electrons closer to its nucleus, or atoms may have same electronegativity and electrons remain at equal distances between nuclei)
Explain the difference between single and double replacement reactions.
single: one single element swaps places with an element in a compound
double: two compounds; one element from each compound swaps places to create two new compounds
Write the formula: Iron (III) oxide
Fe2O3
Explain the octet rule and why it matters
number of electrons required to fill valence shell of all elements (except H and He) and make atoms stable
Explain why subscripts cannot be changed when balancing equations
bc that would be creating a new substance out of thin air i.e. creating matter which breaks the law of conservation of matter
H2 + O2 --> H2O
What is the limiting reagent/reactant given 2 g of H2 and 3 g of O2?
O2
Why is percent yield rarely 100%?
Loss, incomplete reaction
Predict the bond type between magnesium and oxygen and justify your answer.
ionic bc metal and nonmetal
Write a combustion reaction.
CxHy + O2 --> CO2 + H2O
Explain the difference between naming ionic vs covalent compounds (rules + reasoning)
ionic: no greek prefixes; change ending to -ide of second element; include roman numerals for charges of transition metals ONLY
covalent: greek prefixes (except for mono- for first element); change ending to -ide of second element; NO roman numerals
Draw and explain the Lewis structure for CO₂ (include bonding explanation)
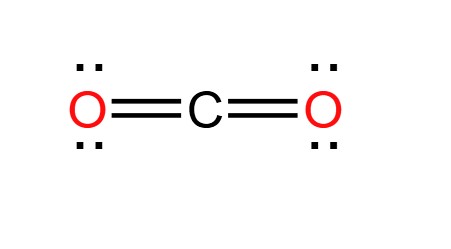 double bonds in order for carbon to fulfill octet rule
double bonds in order for carbon to fulfill octet rule
Balance: C₃H₈ + O₂ → CO₂ + H₂O
C₃H₈ + 5O₂ → 3CO₂ + 4H₂O
H2 + O2 --> H2O
How many grams of H₂O are produced from 10.0 g H₂?
89.1 g H₂O
A chemist is synthesizing ammonia (NH3) by reacting nitrogen gas with hydrogen gas. Based on the amount of starting materials used, the theoretical yield of ammonia for this reaction is calculated to be 25.0 grams.
Upon completing the experiment in the lab, the chemist weighs the final product and finds that only 18.2 grams of ammonia were actually produced.
Calculate the percent yield for this reaction.
72.8%