What is the name of this IONIC compound? Li2S
Lithium Sulfide
What type of reaction is shown here: H2 + O2 --> H2O
Synthesis
Where do we find the molar mass of any element?
The periodic table of elements
What state of matter has the most rigid structure?
Solid
Which side of the reaction arrow can products be found? (left or right)
Right
Formula for IONIC compound containing Calcium and Chlorine
CaCl2
What type of reaction is shown here:
Cu + ZnCl2 --> Zn + CuCl2
Single Replacement
What is the molar mass of Sulfur
32.06g (rounded to 32g)
What does exothermic mean?
The reaction releases energy (heat exits the substance)
True or False: At equilibrium, the amount of reactants equals the amount of products
FALSE
What is the name of this COVALENT compound? C2H4
Dicarbon Tetrahydride
One reactant breaks apart into multiple products
What is the molar mass of the following molecule: Li2O
30g
Li2 = 14
O = 16
Name this phase change: Gas --> Liquid
Condensation
Name the 4 factors that speed up chemical reactions (All 4 are required for points)
What is the formula for the COVALENT compound below?
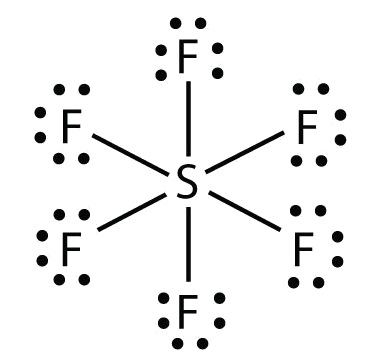
SF6
Oxygen (O2), Carbon Dioxide (CO2), and Water (H2O)
What should the conversion factor look like when converting FROM grams and TO moles?
Grams on the bottom, moles on the top
Is the phase change freezing endothermic or exothermic?
Exothermic (making something colder = removing heat)
Which direction will equilibrium shift if more NaCl is added?
NaOH + HCl → NaCl + H2O + HEAT
Left
What is INCORRECT about this Lewis Dot Diagram? (See image in new tab)
Electrons from the same atom cannot pair. We need two more Fluorine atoms to bond with the remaining Nitrogen electrons
__Na + __MgCl2 --> __Mg + __NaCl
2 Na + 1 MgCl2 --> 1 Mg + 2 NaCl
Convert 2 moles of H2 to grams
4g
(conversion factor --> 2g / 1 mole)
Name a phase change that is endothermic
Melting, Evaporating, or Sublimation
Which direction will the equilibrium shift if this reaction is cooled off?
NaOH + HCl → NaCl + H2O + HEAT
Right