A 54-year-old man is referred to the nephrology clinic to evaluate hypertension. He is currently prescribed maximal doses of fosinopril, labetalol, and chlorthalidone. His office BP, when checked after 5 minutes of quiet rest with a calibrated device, is 142/80 mm Hg with a heart rate of 85/min. His average home BP over the last 7 days has been 145/85 mm Hg. He denies dyspnea, palpitations, headaches, over-the-counter medication use, or recreational drug use.
On physical examination, he appears well. Lungs are clear, and there is no peripheral edema. He has no cardiac murmur or abdominal or carotid bruits.
Laboratory evaluation shows serum creatinine 0.8 mg/dL (reference range, 0.7–1.3), serum potassium 3.9 mEq/L (reference range, 3.5–5.0), and urine sodium 80 mEq/24 hours.
Which ONE of the following is the MOST appropriate next step in management?
A. Measure plasma aldosterone concentration and renin activity
B. Prescribe spironolactone
C. Evaluate for anti-hypertensive medication adherence
D. Recommend dietary sodium reduction
E. Prescribe amlodipine
What is C?
Assessing for medication adherence is the next best step in the management of this patient with apparent treatment-resistant hypertension.
Resistant hypertension (RH) is defined as a clinic BP of ³ 130/80 mm Hg in a patient taking at least three anti-hypertensive agents, including a diuretic, at maximal or maximally tolerated doses. The diagnosis of RH requires the exclusion of pseudo-resistance by verifying uncontrolled hypertension with out-of-office BP measurement and confirmation of adherence to the prescribed medication regimen. Suboptimal adherence is a common cause of uncontrolled hypertension. A 2017 meta-analysis reported a pooled prevalence of nonadherence in patients with apparent treatment-resistant hypertension to be 31.2%. Identification of patients with inadequate adherence can avoid inappropriate intensification of the medication regimen and ensure patients receive the maximum benefit from therapy. Strategies to assess for adherence include patient self-report medication adherence assessment tools, nonthreatening direct questioning, review of pharmacy databases for medication possession and refills, witnessed drug intake, and urine or blood measurement of drug metabolites. In this patient, non-adherence is suggested by his relatively high heart rate despite reported use of labetalol.
A 65-year-old man with metastatic cholangiocarcinoma and type 2 diabetes presents to the emergency department with syncope and polyuria. He reports decreased appetite but no diarrhea or vomiting. He has lost 4 kg over the last 2 weeks and reports dizziness and lightheadedness when standing. He was diagnosed with cholangiocarcinoma 1 year ago and has received cisplatin and gemcitabine since diagnosis, most recently 1 week ago. He reports that he has not been taking his insulin for the last 4 days. Over the last 24 hours, his fluid intake was 1 L, and his urine output was 3.2 L.
His vital signs are as follows:

Urine osm 360
A CT scan of the head is unremarkable.
Which ONE of the following is the MOST likely etiology of his hyponatremia?
A. Glycosuria-mediated osmotic diuresis
B. Syndrome of inappropriate antidiuretic hormone (SIADH)
C. Low solute intake
D. Pseudohyponatremia
E. Renal salt wasting
What Is (E)
This patient with hypovolemic hyponatremia and polyuria most likely has renal salt wasting attributable to cisplatin toxicity.
This patient presents with syncope due to orthostatic hypotension, polyuria, AKI, and hyponatremia. The urine is hypertonic compared with serum, and the urine sodium concentration is elevated despite evidence of volume depletion. Specifically, this hypovolemic patient is losing (125 mEq/L × 3.2 L/day =) 400 mEq/day or 9.2 g/day of sodium in the urine. These findings suggest inappropriate urinary salt wasting. Cisplatin has been associated with renal salt wasting, attributed to several mechanisms. Cisplatin concentrates in proximal tubular cells via the organic cation transporter-2, directly damaging proximal tubular cells and preventing sodium reabsorption. Furthermore, cisplatin binds to and inhibits the basolateral Na+-K+-ATPase and may weakly inhibit the distal sodium epithelial channel, further reducing sodium reabsorption. Low mean arterial pressure leads to secretion of antidiuretic hormone (ADH) with consequent hyponatremia. The treatment of choice is to replete volume with isotonic saline and to maintain a high sodium diet while polyuria persists.
A 24-year-old man presents to the nephrology transplant clinic for evaluation as a potential living kidney donor for his mother who has advanced CKD attributed to a genetic mutation. Family history is also significant for gout in his older brother, and a brain tumor and ESRD requiring dialysis in his maternal grandfather. The patient’s medical history is significant for nocturnal enuresis as a child. He takes no prescription or over-the-counter medication.
His physical examination is unremarkable.
Laboratory data are as follows:
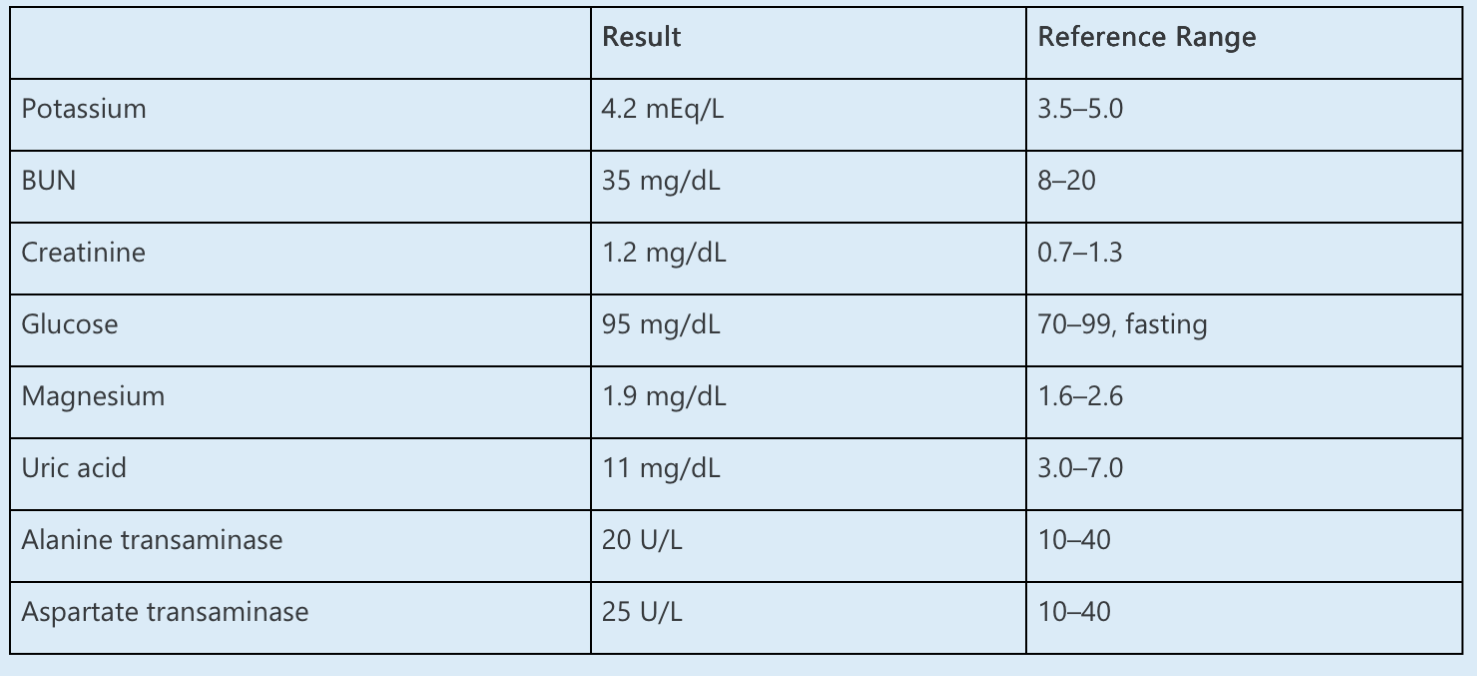
The urine protein-to-creatinine ratio is 0.3 mg/mg (reference range, <0.2) and the fractional excretion of urate is 2%. Urine sediment is notable for rare hyaline casts, no cells, and no crystals.
A kidney ultrasound shows normal-sized kidneys and a 0.5-cm cortical cyst in the left kidney.
Which ONE of the following genes (and gene products) is most likely to be mutated in this family?
A. UMOD (uromodulin)
B. PKD-1 (polycystin-1)
C. VHL (von Hippel-Lindau)
D. HNF-1β (hepatocyte nuclear factor-1β)
E. MUC-1 (mucin-1)
This patient’s presentation with hyperuricemia with low fractional excretion of urate and a family history of kidney disease is virtually pathognomonic for autosomal dominant tubulointerstitial disease (ADTKD) due to a mutation in UMOD, the gene that encodes uromodulin or Tamm-Horsfall protein.
ADTKD refers to a group of monogenic tubulointerstitial diseases characterized by autosomal dominant inheritance and progressive loss of kidney function with a bland urine sediment, minimal proteinuria, and microscopic corticomedullary cysts. Nocturia or enuresis in children may also occur due to loss of renal-concentrating ability. Histopathology is nonspecific and characterized by interstitial fibrosis, tubular atrophy, rare tubular microcysts, and thickening and lamellation of the tubular basement membrane. IF is typically negative for immunoglobulin and complement deposition.
The pathogenic genes and their associated phenotypic correlations are listed below.

A 77-year-old woman is referred for decreased kidney function that was recognized at her annual examination. She had no interval illness or symptoms and was unaware of any kidney problems until informed of her results (below). Her medical history includes hypertension, obesity, sleep apnea, hyperlipidemia, stable coronary artery disease, depression, and osteoarthritis affecting her knees. Eight months ago, she complained of muscle aches, prompting substitution of fenofibrate for simvastatin. Her current medications include amlodipine, fenofibrate, aspirin, fluoxetine, acetaminophen, and glucosamine. She is adherent to her prescription for continuous positive airway pressure (CPAP).
She appears well. BP is 134/67 mm Hg, heart rate is 76/min, and body mass index is 32 kg/m2. Other than trace edema of the legs and degenerative arthritis of her knees, the examination is normal.
Investigations include normal electrolytes and complete blood count, along with the data below:

Urinalysis: specific gravity 1.010, 1+ protein, otherwise negative. Urine sediment: rare hyaline cast.
Kidney ultrasound reveals bilateral 10-cm kidneys with mildly increased echogenicity but no other abnormalities.
Which of the following is the MOST likely explanation for her decline in kidney function?
A. Chronic interstitial nephritis
B. Analgesic nephropathy
C. Hypertensive nephrosclerosis
D. Fenofibrate nephrotoxicity
E. Ischemic nephropathy
What is (D)
This patient has a subacute rise in serum creatinine, bland urine sediment, and minimal proteinuria, as well as chronic exposure to fenofibrate, all of which are most consistent with a diagnosis of fenofibrate nephrotoxicity.
Fenofibrate is a synthetic fibric acid derivative that is utilized for the treatment of dyslipidemia, as it can lower triglycerides and low-density lipoprotein and raise high-density lipoprotein levels. The impact of fenofibrate (and other fibric acid derivatives) on cardiovascular outcomes is unclear, with some studies failing to demonstrate clinical improvement despite a favorable effect on lipid profile. Fenofibrate appears to exert its effect by activating peroxisome proliferator-activated receptor-α (PPAR-α). Most patients appear to tolerate fenofibrate well, but multiple case series have demonstrated that some patients will experience a mild rise in serum creatinine. This rise in creatinine typically occurs after 6 months of therapy or more.
The majority of patients recover to baseline creatinine levels after discontinuation of the drug, but approximately 30% will have persistent CKD. The sediment is bland, and there is usually no significant proteinuria. Kidney biopsies have been infrequently reported in the literature, but when performed, have not demonstrated any specific pathology. One hypothesis regarding the pathophysiology is that fenofibrate (and other fibric acid derivatives) impairs the production of renal vasodilatory prostaglandins. Other studies have suggested that there is direct interference with creatinine excretion or an increase in creatinine production. Fibrates, when combined with statins, may cause rhabdomyolysis that can be associated with AKI, but this is a distinct phenomenon. Kidney transplant recipients treated with calcineurin inhibitors appear to be at particular risk of AKI or CKD with these agents. The propensity to cause CKD appears to be a class effect for fibrates, although there is some suggestion that gemfibrozil may be less problematic than fenofibrate.
A 75-year-old man is referred to the outpatient nephrology clinic to evaluate labile hypertension. The patient has long-standing diabetes mellitus complicated by retinopathy and neuropathy. He has a 10-year history of hypertension, previously well controlled on amlodipine and chlorthalidone, but his medications were discontinued two months ago due to recurrent syncope requiring hospitalization.
On physical examination, supine BP is 184/110 mm Hg, and pulse is 78/min. Standing BP after 3 minutes is 100/80 mm Hg, and pulse is 85/min and is accompanied by lightheadedness. Home BP readings confirm these findings. The patient has previously tried wearing compression stockings, an abdominal binder, and drinking cold water and crossing his legs before standing with little benefit.
Which of the following is the MOST appropriate medication for managing supine hypertension in this patient?
A. Chlorthalidone
B. Amiloride
C. Clonidine
D. Amlodipine
E. Spironolactone
What is (C)?
Supine hypertension is common in patients with neurogenic orthostatic hypotension. In addition to lifestyle changes, short-acting drugs such as nitrates or clonidine can be administered, typically at bedtime, to control supine BP in patients with severe supine hypertension.
Orthostatic hypotension (OH) is defined as a sustained reduction of systolic BP (SBP) of at least 20 mm Hg or diastolic blood BP (DBP) of at least 10 mm Hg within 3 minutes of standing. The evaluation and management of OH entails removing or modifying offending drugs and excluding adrenal insufficiency and volume depletion secondary to gastrointestinal or renal fluid losses. Neurogenic causes should be suspected once drug- and volume-associated OH have been ruled out. Common causes of neurogenic OH include α-synucleopathies (i.e., Parkinson disease, Lewy body dementia, multisystem atrophy) or peripheral neuropathies (i.e., diabetes mellitus, amyloidosis, hereditary autonomic neuropathies, Guillain-Barre syndrome).
In normal subjects, on standing, about 800–1000 mL of blood pools in the lower body veins, and approximately 10% of the fluid shifts from the intravascular space to the interstitial space because of increased hydrostatic pressure. The decrease in venous return and subsequent reduction in stroke volume results in decreased baroreflex activation, leading to increased sympathetic tone and decreased parasympathetic tone. The sympathetic tone increases peripheral vascular resistance, and any disruption of these pathways (central or peripheral neurological disorders) may result in neurogenic OH. Management involves patient education, including strategies to mitigate OH and removing or modifying drugs that can worsen OH. Long-acting hypertensive medications should be discontinued in these patients, especially those who are symptomatic.
A 60-year-old man with a past medical history of hypertension and hyperlipidemia presents to his primary care physician to evaluate fatigue and low back pain. He was last seen two years ago. His home medications include atorvastatin, amlodipine, and chlorthalidone. He has also been taking over-the-counter ibuprofen three times daily for the last month.
On examination, his BP is 160/80 mm Hg, with a heart rate of 80/min. He is in no acute distress and appears his stated age. There is no evidence of a cardiac murmur or rub. He has no peripheral edema. He has no point tenderness on palpation of his back and no rashes or lesions noted.
Laboratory data are as follows:

Which ONE of the following is the MOST likely cause of this patient’s low anion gap?
A. Paraproteinemia
B. Hypertriglyceridemia
C. Ibuprofen consumption
D. Laboratory error
E. Chlorthalidone
What is (A)
The most likely explanation for this patient’s low anion gap is unmeasured, cationic light chains (paraproteins) suggesting underlying multiple myeloma (MM).
The anion gap, calculated as serum Na – (Cl + HCO3), is most commonly used to evaluate for unmeasured anions to further characterize a metabolic acidosis. The normal anion gap varies by local laboratory but is usually in the range of 7–13 mEq/L, or approximately two to three times the albumin (normal, ~4) measured in g/dL. This patient’s serum anion gap is 139 – (110 + 28) = 1 mEq/L.
This patient presents with symptoms and laboratory findings concerning for MM, including fatigue, back pain, worsening kidney function, hypercalcemia, and an elevated protein gap (total protein – albumin >4 g/dL), which suggests unmeasured serum proteins such as paraproteins. Patients with plasma cell dyscrasias may accumulate monoclonal immunoglobulins that are typically cationic and occur in the serum as chloride salts, causing elevated serum chloride and a low or even negative anion gap.
A 32-year-old man with sickle cell disease and type 1 diabetes presents to the emergency department with 3 days of diffuse pain, fever, chest pain, and shortness of breath. He is intubated for respiratory distress and admitted to the ICU. His current medications include insulin, ceftriaxone, azithromycin, and morphine. He is receiving tube feeding with 250 mL of free water flush every 4 hours via nasogastric tube. Urine output is 3.2 L/24 h.
On examination, his temperature is 38.1°C, BP is 100/50 mm Hg, heart rate is 124/min, and oxygen saturation is 94% while ventilated with 40% oxygen. He is sedated, with coarse breath sounds and tachycardia. He has an indwelling urinary catheter draining abundant, clear yellow urine.
Laboratory data are as follows:

Which ONE of the following is the MOST likely etiology of his polyuria?
A. Excess water intake
B. Hypocalcemia
C. Hyperglycemia
D. Sickle cell disease
E. Excess protein intake
What is (D)
The most likely cause of polyuria is sickle cell disease (SCD).
SCD commonly causes urinary concentrating defects (hyposthenuria). As hemoglobin traverses the renal medulla, the relative hypoxia within the vasa recta leads to increased sickling of the RBC and vascular occlusion followed by capillary rarefaction and distal tubular dysfunction. This can cause decreased sodium reabsorption in the renal medulla at the loop of Henle and the inability to generate the normal medullary concentration gradient required for maximal water reabsorption. Moreover, distal tubular dysfunction may cause an inability to absorb water appropriately (concentrating defect, nephrogenic diabetes insipidus). Early manifestations of urinary concentrating defects include nocturnal enuresis, which is reported to occur in 60% of children with SCD. Other manifestations of distal tubule failure include distal renal tubular acidosis and impaired potassium excretion. One consequence of this pathophysiology is proximal tubular hypertrophy, which can result in increased creatinine secretion at the proximal tubule and overestimation of creatinine-based eGFR in SCD.
A 75-year-old woman who resides in a skilled nursing facility due to chronic obstructive pulmonary disease and frailty (body weight, 50 kg) presents with fever and hypotension without an apparent source of infection. An infectious disease consultant recommends empiric antibiotic coverage with vancomycin and piperacillin-tazobactam pending culture results.
She is admitted to the ICU, where she requires mechanical ventilation. In addition to the antibiotics, her medications include propofol, omeprazole, bronchodilators, acetaminophen, and low-molecular weight heparin to prevent venous thrombosis. Over the ensuing 96 hours, her total fluid intake is 13 L, and her total urine output is 3 L. Her serum creatinine increases to 1.1 mg/dL (baseline, 0.7; reference range, 0.5–1.1). Liver function tests are normal except for a reduced serum albumin.
Urinalysis via a urinary catheter-derived specimen reveals specific gravity 1.010, pH 6, and trace blood and protein. The sediment contains 1–3 RBCs/hpf, 1–3 WBCs/hpf, and occasional granular casts.
Which of the following studies is MOST likely to yield an explanation for the acute decline in kidney function in this patient?
A. Ultrasound of the kidneys and bladder
B. Serum creatine kinase level
C. Urine sodium and creatinine levels
D. Vancomycin trough level
E. Urine eosinophil stain
What is (D)
A vancomycin trough level should be obtained in this patient with AKI.
The first step in the evaluation of the AKI in this patient is to recognize the severity of the problem. In this case, marked positive fluid balance has led to expansion of the volume of distribution of serum creatinine, the principal biomarker used to identify AKI. This dilution blunts the rise in creatinine and may “mask” AKI, delaying recognition and contributing to a poor outcome. The creatinine concentration can be corrected for the degree of volume expansion by multiplying by a correction factor that accounts for expansion of total body water:
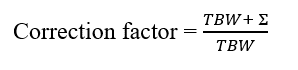
TBW represents total body water, estimated as 0.5 × admission weight, and ∑ represents the cumulative daily fluid balance:
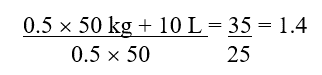
In this case, the correction factor is 1.4, so 1.4 × 1.2 mg/dL results in a corrected creatinine of 1.68 mg/dL, more than double her admission value and consistent with stage 2 AK
A 56-year-old man presents to the outpatient clinic to establish care. His past medical history is notable for hyperlipidemia, for which he takes pravastatin. He feels well and does not use tobacco or alcohol. He reports that he often eats take-out food and that his lifestyle is sedentary.
His physical examination is unremarkable with the exception of a BP of 158/97 mm Hg and a body mass index of 30 kg/m2. A basic metabolic panel and complete blood cell count are normal, and a fasting lipid panel reveals total cholesterol 180 mg/dL (reference range, <200) and HDL 32 mg/dL (reference range, low <40 mg/dL). A UA is unremarkable.
An EKG shows normal sinus rhythm with left axis deviation.
He is prescribed a BP monitor and given instructions to measure his BP at home twice daily for 2 weeks. He returns 3 weeks later for follow-up, and his home BP average is 156/94 mm Hg. He is educated on lifestyle interventions to lower BP, including a low-sodium, heart-healthy diet and regular physical activity, and he appears motivated to make the suggested changes.
Which of the following is the most appropriate NEXT STEP in management?
A. Prescribe benazepril-amlodipine
B. Prescribe hydrochlorothiazide
C. Prescribe atenolol
D. Measure plasma renin activity and aldosterone level
E. Reassess BP 3 months after intensive lifestyle changes
What is (A)
This patient with newly diagnosed stage 2 hypertension should be initiated on two antihypertensive medications, such as benazepril-amlodipine.
This patient has hypertension, which has been confirmed with out-of-office BP measurements, and he has other cardiovascular disease risk factors, including hyperlipidemia and obesity, which lead to an estimated 10-year atherosclerotic cardiovascular disease risk of 10%. Based on cardiovascular risk, his target BP is <130/80 mm Hg. Prompt evaluation and treatment of stage 2 hypertension with a combination of drug and nonpharmacological therapy are important due to the high risk of cardiovascular events. Initiation of antihypertensive drug therapy with two first-line agents of different classes, either as separate agents or in fixed-dose combination, is recommended in adults with stage 2 hypertension and an average BP >20/10 mm Hg above their BP target. Evidence favoring this approach comes from studies using fixed-dose combinations, which showed greater BP reduction compared with single agents and better adherence. Therefore, the best answer is to start benazepril-amlodipine.
Which process plays a role in maintenance of metabolic alkalosis rather than the generation of metabolic alkalosis?
A. Decreased glomerular filtration rate
B. Metabolism of citrate
C. Parenteral high-dose penicillin salt administration
D. Overnutrition in a malnourished patient
What is (A)
Answers B–D all describe mechanisms that contribute to the generation of alkalosis; thus only answer A describes a mechanism that maintains metabolic alkalosis.
Generation phase of metabolic alkalosis can be broken down into a loss of acid or a gain in bicarbonate. Examples of loss of acid include the loss of HCl with vomiting or nasogastric suction, loss of excessive chloride in sweat, intracellular H+ transfer such as in hypokalemia, or renal loss in H+ from either decreased Na+, K+, or Cl− reabsorption or increased sodium reabsorption with hypervolemia and hypertension. Bicarbonate concentration can increase from
exogenous alkali (e.g., antacids, sodium bicarbonate, or milk alkali syndrome), medications (e.g.,nonabsorbable anions), metabolism of bicarbonate precursors (e.g., citrate, acetate, or lactate),or overnutrition resulting in refeeding syndrome, causing increased formation of ketoacids, which break down into bicarbonate.
A 21-year-old man is evaluated in the emergency department for a possible concussion during a football game. He has no known medical history but notes having episodes of muscular weakness associated with some lightheadedness and headache over the last several months.
His physical examination is remarkable for a BP of 170/98 mm Hg (similar in both arms), pulse of 68/min, and body mass index of 20 kg/m2. He is alert and oriented with no deficits on neurological exam. The remainder of the examination is normal.
Laboratory data reveal the following:

Complete blood count, creatine kinase level, and liver function tests are normal. EKG demonstrates mildly peaked T waves. CT scan of the brain demonstrates no acute intracranial abnormality.
Treatment with fludrocortisone does not increase urinary potassium excretion; however, when fludrocortisone is combined with isotonic sodium bicarbonate in doses large enough to sustain a serum bicarbonate level greater than 25 mEq/L and a urinary pH greater than 8, then urinary potassium concentration increases to 80 mEq/L.
Which one of the following is the MOST likely cause of this patient’s laboratory abnormalities?
A.
Hyporeninemic hypoaldosteronism
B.
A mutation in either WNK1 or WNK4 (with no lysine) kinases in the distal convoluted tubule
C.
Primary adrenal insufficiency (Addison disease)
D.
A point mutation in the gene for the α-subunit of the skeletal muscle cell sodium channel
E.
A loss of function mutation in the gene encoding the principal cell mineralocorticoid receptor
What is (B)
The combination of hyperkalemia, hypertension, and mild metabolic acidosis in a young individual is characteristic of pseudohypoaldosteronism type II (Gordon syndrome), which is caused by either a gain of function mutation in WNK1 (with no lysine kinase-1) or a loss of function mutation in WNK4. Both abnormalities result in increased function of the NaCl cotransporter and the epithelial Na channel, as well as profound inhibition of the renal outer medullary potassium channel (ROMK) channels in the cortical collecting duct. The net effect is decreased Na and K excretion. In addition, there is likely a reduction in the luminal electrical gradient secondary to a chloride shunt (increase in paracellular chloride permeability leading to electroneutral sodium chloride reabsorption). Renin and aldosterone are typically suppressed due to volume expansion from the enhanced sodium reabsorption at the distal nephron. Administration of fludrocortisone alone will not substantially increase the rate of potassium excretion given that there is still a marked inhibition of ROMK. However, when combined with sodium bicarbonate administration sufficient to cause bicarbonaturia, there will be an increase in K trapping in the lumen from more proximal sites, thereby resulting in kaliuresis.
A 60-year-old man with hepatosplenic T-cell lymphoma presents to the emergency department with 1 month of progressive fatigue and muscle weakness. For the last 3 months, he has been undergoing chemotherapy with ifosfamide, carboplatin, and etoposide. His last treatment was 1 week ago, and since that time, he has had nausea, vomiting, diarrhea, and poor oral intake.
On examination, BP is 104/58 mm Hg, and heart rate is 96/min. His lungs are clear, and there is no lower extremity edema. He has diminished strength in all extremities. The remainder of the examination is unremarkable.
Laboratory data are as follows:

Which ONE of the following is the MOST likely etiology of his electrolyte abnormalities?
A. Ifosfamide
B. Malnutrition
C. Etoposide
D. Carboplatin
E. Diarrhea
What is (A)
This patient with hypokalemia, hypophosphatemia, hypouricemia, and non-anion gap metabolic acidosis most likely has generalized proximal tubule dysfunction from ifosfamide toxicity.
Electrolyte abnormalities are common complications of chemotherapy. Ifosfamide may adversely affect kidney function in several ways, including AKI due to acute tubular injury, CKD, and proximal tubulopathy (Fanconi syndrome). Ifosfamide is transported via the apical organic cation transporter OCT-2 from the urinary filtrate into proximal tubule cells. Inside the cell, the drug is metabolized into chloroacetaldehyde, where it can accumulate and disrupt oxidative phosphorylation. AKI occurs in up to 60% of patients receiving ifosfamide, and up to 30% progress to CKD. Risk factors include cumulative dose, concurrent cisplatin therapy, and nephrectomy. Manifestations of Fanconi syndrome include hypokalemia, hypophosphatemia, hypouricemia, aminoaciduria, non-anion gap metabolic acidosis, and normoglycemic glycosuria. The diagnosis of Fanconi syndrome can be confirmed by calculating the fractional excretion of each electrolyte, which should be elevated.
You are a member of your organization’s quality improvement committee, and the committee’s current focus is on how to improve rates of BP control across the organization. The decision is made to focus on specific interventions at the level of individual practices.
Which of the following single interventions is likely to yield the greatest improvement in rates of BP control?
A. Implement an automated texting system reminding patients to check their BP at home
B. Implement a guideline-based protocol that allows clinic nurses to titrate medication doses under the final approval of the ordering physician, and order laboratory tests and follow-up on the basis of patient BP readings
C. Implement a new artificial intelligence software that reviews the patient’s electronic medical record to identify patterns of high BP readings at office visits
D. Implement patient education visits for nurses to provide patient education on home BP measurement, antihypertensive medications, and lifestyle modification
What is (B)
Although multiple interventions have been shown to improve patient BP control, use of a multidisciplinary team with physician-supervised protocols that allow medication titration have been shown to produce the greatest degree of BP lowering and reduce time needed to achieve optimal BP.
A 63-year-old female presents to the emergency room obtunded and unable to give a history. Vital signs: temperature: 98.8, pulse: 112, Blood pressure: 92/64, pulse oximeter: 100% on room air. Physical exam reveals dry mucous membranes and patient unable to respond to commands or answer questions appropriately, but she does withdraw from pain in all extremities. Laboratory data

Her previous baseline creatinine was 0.9 mg/dl.
Urine toxicology was negative. Finger stick glucose was within normal limits. The head CT scan was normal. Urinalysis revealed a pH of 6.8. Spot urine electrolytes: urine K+ 12 mEq/L, Cl- 32 mEq/L, and sodium 52 mEq/L.
Based on these findings, what is the most likely cause for this patient’s metabolic alkalosis?
A. Vomiting.
B. Bartter syndrome.
C. Milk Alkali Syndrome.
D. Liddle’s Syndrome (pseudohyperaldosteronism).
E. Nonabsorbable anion.
What is (C)
The patient demonstrates metabolic alkalosis in the setting of AKI and hypercalcemia, which is very suggestive of milk alkali syndrome. This is further verified by a high urine pH. The high urine sodium is suggestive of the ongoing natriuresis associated with
hypercalcemia. Urine chloride is elevated to maintain electroneutrality given the large number of cations present in the tubular lumen.
A 50-year-old man with a past medical history of hypertension and obesity presents to his primary care physician with fatigue, constipation, and leg cramping, the last of which is worse at night. His medications include amlodipine and lisinopril. He reports a 50-pack-year history of cigarette smoking but has recently been transitioning to chewing tobacco in preparation for smoking cessation.
On examination, his BP is 176/105 mm Hg and his heart rate is 65/min. His cardiopulmonary examination is unremarkable, and he has mild, bilateral lower extremity edema. Muscle fasciculations are seen in his calves.
Laboratory data are as follows:

He has no prior history of hypokalemia
EKG shows a prolonged PR interval and flattened T waves.
Which ONE of the following is the pathophysiologic mechanism causing hypertension and hypokalemia?
A. Mutation in genes encoding the epithelial sodium channel (ENaC) subunits
B. Intracellular potassium shift due to metabolic alkalosis
C. Reduced activity of 11β-hydroxysteroid dehydrogenase
D. Mutation of distal convoluted tubule WNK1 gene
E. Malnutrition and decreased potassium intake
What is (C)
This patient, with evidence of the syndrome of apparent mineralocorticoid excess (SAME), likely has reduced activity of 11β-hydroxysteroid dehydrogenase.
This patient has symptomatic hypokalemia, metabolic alkalosis, hypertension, and edema. These findings are often associated with overactivation of the renin-angiotensin-aldosterone system (RAAS), usually mediated by excess renin or aldosterone. However, his renin and aldosterone levels are low, suggesting that RAAS is suppressed, consistent with SAME.
Licorice is derived from the Glycyrrhiza glabra plant, and one of its components, glycyrrhizic acid, is metabolized to glycyrrhetinic acid, known to cause SAME by inhibition of the 11β-hydroxysteroid dehydrogenase type 2 enzyme. In normal physiologic states, 11β-hydroxysteroid dehydrogenase converts cortisol to the inactive cortisone, which is unable to activate the mineralocorticoid receptor. However, if this enzyme is inhibited, cortisol persists to stimulate the receptor independent of aldosterone. The resultant hypertension leads to suppression of the RAAS axis, and the excess activation of the mineralocorticoid receptor leads to hypokalemia.
Both black licorice candy and chewing tobacco may contain licorice root or extract, and their ingestion may cause SAME, as is illustrated in this case.
Additional evaluation for SAME includes a 24-hour urine collection for cortisol and cortisone. If the cortisone-to-cortisol ratio is <1 (reference range, >2:1), then SAME is likely.
A 65-year-old woman with a history of ESRD, hypertension, coronary artery disease, peripheral vascular disease, and unipolar major depression is seen during routine HD. During a recent evaluation by her psychiatrist, she was diagnosed with a major depressive episode. She reports that over the last 6 weeks, she has been experiencing depressed mood. She feels hopeless and sad nearly every day, shares feelings of worthlessness, and finds very little interest or pleasure in activities that she used to enjoy. She also describes insomnia, unintentional weight gain, severe fatigue, and poor concentration, such that she no longer spends time with family or friends.
On examination, she appears tearful, tired, and poorly attentive. BP is 104/80 mm Hg, and heart rate is 66/min.
Which ONE of the following is the MOST appropriate pharmacotherapy for this patient?
A. Paroxetine
B. Citalopram
C. Escitalopram
D. Imipramine
E. Phenelzine
What is (A)
This patient with ESRD receiving HD and major depression should be prescribed a selective serotonin reuptake inhibitor (SSRI) such as paroxetine.
For patients receiving maintenance HD and treatment for unipolar major depression, evidence is limited regarding the relative efficacy of combination psychotherapy plus pharmacotherapy (antidepressants); psychotherapy alone; or pharmacotherapy alone. Treatment options may be largely guided by patient preference and access to these resources.
SSRIs are generally recommended as first-line agents for patients receiving dialysis due to established efficacy and relatively favorable side effect profile. Sertraline, paroxetine, and fluoxetine have been well studied and are some of the most recommended SSRIs. Dosing adjustment for paroxetine is needed in patients with a creatinine clearance <30 mL/min. Citalopram and escitalopram should be avoided in patients with ESRD due to the potential for QT prolongation and/or increased risk of sudden cardiac death.
Selective norepinephrine reuptake inhibitors (SNRIs) and atypical antidepressants (bupropion, mirtazapine) have also demonstrated efficacy for the treatment of depression in patients with ESRD and carry additional properties that may be helpful for concurrent therapy of neuropathic pain, sleep disorders, and appetite stimulation, respectively.
A 53-year-old man with a history of CKD stage 3b, hypertension, and a left lower extremity deep vein thrombosis 3 months ago is evaluated in clinic for follow-up. His current medications include apixaban, losartan, spironolactone, empagliflozin, and chlorthalidone.
His physical examination is notable for a BP of 172/106 mm Hg and a heart rate of 78/min. Body mass index is 26 kg/m2. His cardiovascular examination is unremarkable with no murmurs or extra heart sounds. The abdomen is soft, without bruits. He has no peripheral edema.
Laboratory data are as follows:

A UA was significant for 1+ protein. A spot urine albumin-to-creatinine ratio was 150 mg/g (reference range, <30).
After confirming adherence to his medication regimen, he is prescribed amlodipine 10 mg daily. He returns to the clinic 3 months later and his BP is 156/88 mm Hg. He reports no complaints.
A repeated UA shows 2+ protein. A spot urine albumin-to-creatinine ratio is now 654 mg/g.
Which ONE of the following is the MOST appropriate next step in management?
A. Change amlodipine to diltiazem
B. Prescribe semaglutide
C. Increase spironolactone dose
D. Change spironolactone to finerenone
E. Perform kidney biopsy
What is (A)
This patient, who developed worsening proteinuria after initiating amlodipine, should have amlodipine changed to diltiazem.
Dihydropyridine calcium channel blockers such as amlodipine can exacerbate proteinuria by preferentially dilating renal afferent arterioles, leading to increased intraglomerular pressure and worsened proteinuria. Non-dihydropyridine calcium channel blockers, such as diltiazem or verapamil, have antiproteinuric effects that may make them more appropriate agents for patients with hypertension and significant proteinuria.
A 72-year-old patient with end-stage renal disease secondary to type 2 diabetes, gout, hypertension, secondary hyperparathyroidism, coronary artery disease, and gastroesophageal reflux comes in for his routine dialysis. He has been on dialysis for the past 3 years. It has been noted that he has been alkalotic on several occasions, and repeat monthly labs were checked. Patient is currently on amlodipine, atenolol, sevelamer hydrochloride, simvastatin, and erythropoietin. He reports no recent hospital admissions. His effluent dialysate bicarbonate concentration has been previously decreased to 25 mEq/L, and repeat monthly labs were checked. His estimated dry weight, erythropoietin dose, and hemoglobin have been stable over the past few months. He does not have any urine output.
Vitals: Blood pressure 152/98, pulse: 64. blood flow rate: 400 ml/min.Dialysate flow rate: 800 ml/min
Predialysis labs
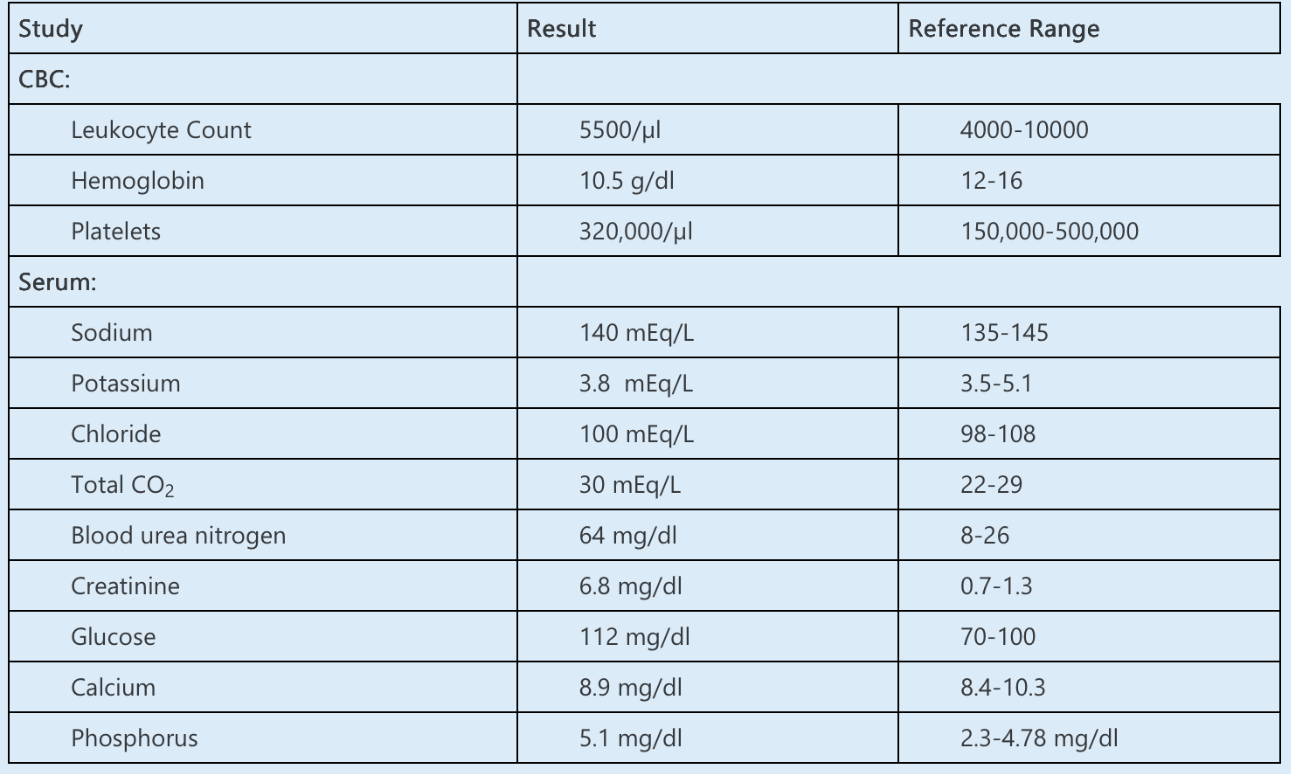
kT/V: 1.5.
What can possibly contribute to this patient’s elevated bicarbonate?
A. Sevelamer hydrochloride use.
B. Licorice use.
C. Obstructive sleep apnea.
D. Pica.
What is (D)
The patient has persistent elevation in bicarbonate. Answer D is most correct. This patient should be screened for other forms of exogenous alkali load including sodium bicarbonate use from either pica or treatment of medical conditions such as gastroesophageal reflux disease or crack cocaine use. His hemoglobin being stable suggests against any massive transfusions.
A 38-year-old woman with a history of migraine headaches, acne vulgaris, and nephrolithiasis presents to the emergency department with six weeks of progressive generalized weakness and fatigue. She also reports intermittent diarrhea and poor appetite. Her current prescription medications include topiramate and spironolactone. Her over-the-counter medications include acetaminophen as needed for headaches.
On examination, her BP is 101/65 mm Hg and her heart rate is 108/min. The remainder of her examination is unremarkable.
Laboratory data are as follows:
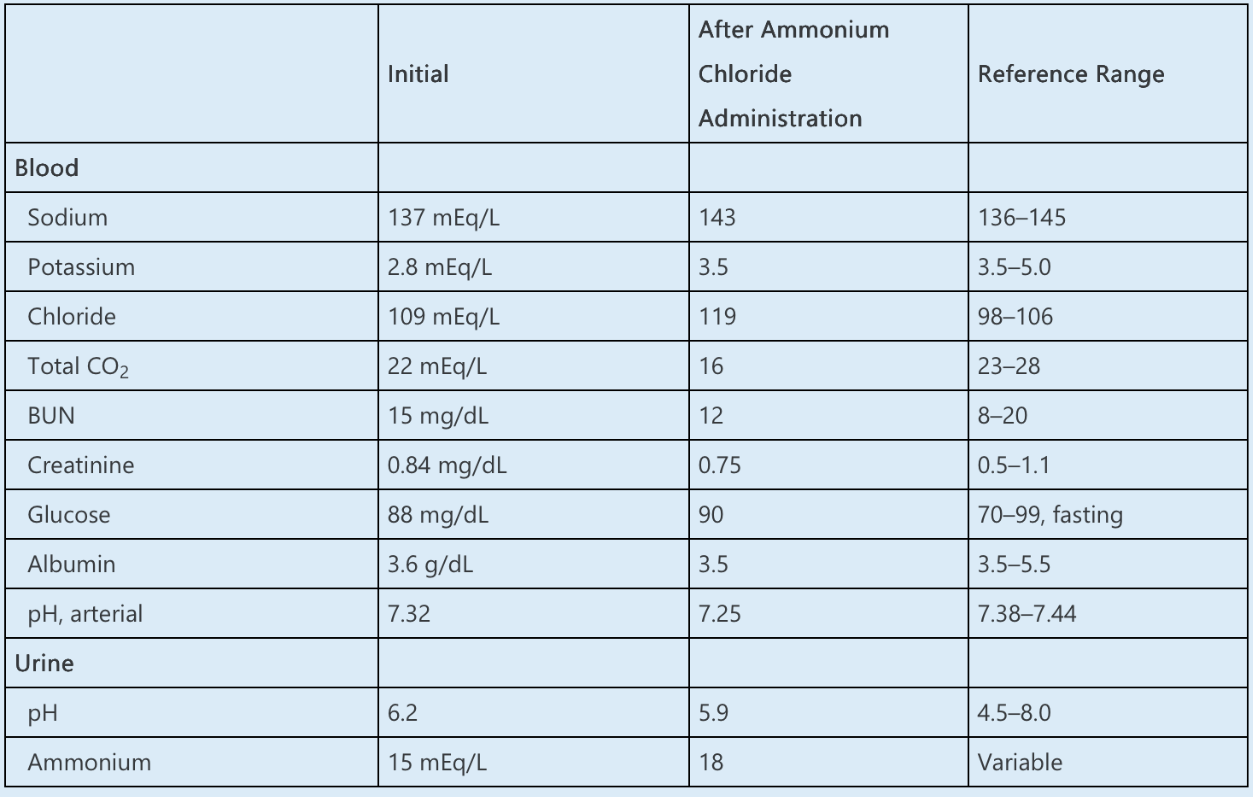
Which ONE of the following is the MOST likely cause of this patient’s acidemia?
A. Proximal renal tubular acidosis
B. Pyroglutamic acidosis
C. Spironolactone
D. Distal renal tubular acidosis
E. Diarrhea
What is (D)
The most likely etiology of a normal anion gap acidosis with an inappropriately elevated urinary pH and decreased ammonium excretion is distal renal tubular acidosis (RTA).
A normal anion gap acidosis is caused by a nonrenal loss of bicarbonate, impaired renal acid excretion, or dilution of the blood by administration of IV fluids that do not contain bicarbonate. In this patient with acidemia and a normal anion gap, the first step is to distinguish between gastrointestinal (GI) and renal etiologies. Urinary ammonium excretion increases in diarrhea-induced acidosis (up to 200–300 mmol/day), whereas a low value, as in this case, suggests a defect in renal acid excretion, such as RTA.
This patient has several possible reasons for RTA. To determine the most likely etiology, it is necessary to distinguish between proximal and distal RTA. In distal RTA, there is impaired acid excretion (defect in H+ secretion), which results in an inability to reduce the urinary pH to <5.3 in the setting of acidemia. In proximal RTA, there is defective bicarbonate reabsorption in the proximal tubule, but distal acidification remains intact. Thus, in the setting of acidemia, urinary pH will be appropriately low in proximal RTA but inappropriately elevated in distal RTA, as in this case.
Loading with ammonium chloride (NH4Cl) can distinguish between proximal and distal RTA. Administration of an acidifying salt should enhance H+ and K+secretion in the cortical collecting tubule. If urine pH remains >5.3 despite this acid load, distal RTA is confirmed, whereas in proximal RTA, NH4Cl loading should enhance preserved distal acidification mechanisms, resulting in a urine pH <5.3.
A 59-year-old man with no known medical history is brought to the emergency department with fever, headache, and confusion. He is diagnosed with HIV and cryptococcal meningitis. He has a prolonged hospitalization that is complicated by seizures, hypertension, diarrhea, and urinary tract obstruction from benign prostatic hyperplasia. His current medications include amphotericin B, tenofovir, emtricitabine, bictegravir, carbamazepine, topiramate, lisinopril, and tamsulosin.
After 4 weeks, he develops acidemia and electrolyte disturbances, for which he receives sodium bicarbonate IV.
Laboratory data are as follows:

Which ONE of the following is the MOST likely cause of this patient’s acidosis?
A. Tenofovir
B. Amphotericin B
C. Diarrhea
D. Topiramate
E. Urinary obstruction
What is (B)
The most likely etiology of a normal anion gap acidosis with an inappropriately elevated urinary pH and decreased ammonium excretion is distal renal tubular acidosis (RTA), in this case caused by amphotericin B.
A normal anion gap acidosis is caused by non-renal loss of bicarbonate, impaired renal acid excretion, or dilution of the blood by administration of IV fluids that do not contain bicarbonate (or potential bicarbonate). In this patient with acidemia and a normal anion gap, the first step is to distinguish between gastrointestinal (GI) and renal etiologies. Urinary ammonium excretion increases in diarrhea-induced acidosis (up to 200–300 mEq/d), whereas a low value, as in this case, suggests an RTA. The urine anion gap, frequently used to estimate urinary ammonium excretion, has been shown in recent studies to be unreliable.
This patient has several potential reasons for an RTA. To determine the most likely etiology, it is necessary to distinguish between proximal RTA and distal RTA. In distal RTA, there is impaired acid excretion (defect in H+ secretion) that results in an inability to reduce the urinary pH to <5.5 in the setting of acidemia. In proximal RTA, there is defective bicarbonate reabsorption in the proximal tubule, but distal acidification remains intact. Thus, in the setting of acidemia, urinary pH will be appropriately low in proximal RTA. These relationships are only approximate in actual practice, as urine ammonium production and H+ secretion rates may differ depending on the acuity of the acidosis, confusing the relationship between urine pH and urine ammonium production.
Infusion of sodium bicarbonate can aid with diagnosis of proximal versus distal RTA. In proximal RTA, infusion of sufficient sodium bicarbonate to raise the serum bicarbonate concentration level to 18–20 mEq/L will result in an increase in urine pH to above 7.5 (as the reabsorptive threshold for bicarbonate is exceeded), whereas urine pH will remain stable in distal RTA. Furthermore, the fractional excretion of bicarbonate will exceed 15%–20% in proximal RTA, whereas remaining <3% for distal RTA. This patient’s urine pH is >5.5 in the context of acidemia with little change after sodium bicarbonate infusion, leading to a diagnosis of distal RTA. Amphotericin B can cause an acquired distal RTA by increasing membrane permeability in the distal tubule, leading to back-diffusion of secreted H+ ions from the lumen into the distal tubular cell and impairing urinary acidification.