What are coenzymes?
Where would you find coenzymes?
Coenzymes can be either inorganic or organic cofactors that are additional factors that can work w/ proteins to provide a broader range of chemistry at the active site, allowing enzymes to catalyze a wider ranger of reactions.
Both organic and inorganic cofactors are often derived from vitamins- hence the dietary requirement
*More info on slide 2, lecture 16*
What are carbohydrates? List 3 types of carbohydrates.
5pt. bonus if know basic formula of carbohydrates.
5pt. bonus how are di/tri/polysaccharides linked together?
Carbohydrates are sugars & polymers of sugars. They vary in carbon #, position of carbonyl group, and arrangement of atoms.
3 types: Monosaccharides(single sugar)(monomer), Disaccharides(two sugar), and Polysaccharides(multiple sugars)(polymer).
di/tri/polysaccharides joined by glycosidic bonds between alpha glucose carbons between 1' and 4' carbons.
basic formula: (CH2O)n
Exam hint: these usually end in -ose
What is ATP?
Adenosine triphosphate(ATP) is the energy "currency" of the cell. It is a high free energy molecule. And most of the potential energy comes from the three phosphate groups that are negatively charged and crowded together.
How do Biological endergonic reactions occur?
Biological systems ned to drive endergonic reactions by coupling them with exergonic reactions. The overall delta G of coupled reactions is negative.
What is a reduction-oxidation(redox)reaction? What do they drive?
Redox reactions are chemical reactions that involve electron transfer. Redox reactions drive ATP formation.
List examples of inorganic and organic cofactors.
Inorganic: also directly interact w/ substrates to lower energy of activation.
Examples are metal ions like: Zn2+, Mg2+, Fe2+, Cu2+, etc.
Organic: NAD+ found in Niacin, FAD found in Riboflavin, Coenzyme A(CoA) found in Pantothenic acid, Coenzyme B-12 found in vitamin B-12, etc.
*More info on slide 4 lec. 16*
What are stereoisomers? Being stereoisomers of each other, how do alpha and Beta glucose differ?
Stereoisomers are molecules with the SAME molecular formula and sequence of bonded atoms(constitution), but differ in their spacial orientations of their atoms.
They differ in positioning of the ring oxygen and 1' OH group, despite subtle difference they behave very differently in biological settings.
Define Catabolism and Anabolism.
Catabolism: The breakdown of macromolecules(e.g. Carbohydrates, Proteins, Fats, nucleic acids) to subunits(Sugars, Amino Acids, Fatty Acids, nucleotides) often to extract energy.
Where in cells have the most important source of chemical potential energy?
What is the amount of potential energy based on?
The electrons are the most important source of chemical potential energy.
Electrons closer to negative charges and farther from positive charges have higher potential energy.
What is it called when an atom or molecule gains an electron? What is it when it loses an electron? What are the agents for each?
When an atom or molecule gains an electron it is REDUCED and is the Oxidizing agent.
When an atom or molecule loses an electron it is OXIDIZIED and is the reducting agent.
*Redox events are always coupled, if one atom loses an electron, another has to gain it.*
What type of inhibitors are most drugs?
Competitive inhibitors.
Which type of glucose is easy to digest? Why?
Alpha-glucose polymers(e.g. starch) are east to digest, while Beta-glucose polymers(e.g. cellulose) are hard to digest. This is because beta-glucose polymers like cellulose readily line up together and form many H-bonds, making insoluble bundles, with very little available for enzymatic hydrolysis.
What is the Metabolism? List the 3 parts of the metabolism in order.
The metabolism is the sum of all chemical reactions in a cell. Metabolic Reactions involve Energy Exchange.
The Three parts of the metabolism in order are: Glycolysis, Kreb's Cycle/Citric Acid Cycle, and Electron Transport & Oxidative Phosphorylation
What is the overall equation for Aerobic Cellular Respiration?
C6H12O6 + 6O2 --> 6CO2 + 6H2O + energy
What are the 4 key energy-carrying molecules in biological systems?
Glucose, ATP, NADH, and FADH2
Need to know the Vmax. More context on Slide 17
800pts.
Where is the extracellular Matrix and what is it comprised of? Go into detail of the components.
10pt bonus if can list some examples of each of the components.
The extracellular Matrix is located outside of the anima plasma membrane; and is composed of glycoproteins, polysaccharides, and proteoglycans.
Glycoproteins: proteins w/ some attatched carbohydrate groups. Examples: laminin, type IV collagen, nidogen.
Proteoglycans: long polysaccharide chains linked to a specific core proteins: perlecan, aggrecan
Polysaccharides: long chain polysaccharides. Ex. hyaluronan
Is ATP Hydrolysis Exergonic or Endergonic? What is it's delta G?
Exergonic, with a delta G of -7.3
1.What are the free energy states of electrons in polar and non-polar covalent bonds?
2.What happens if you change bonds from non polar covalent to polar covalent?
3.What happens when going from high free energy of np covalent bonds to low free energy of polar bonds?

1. The electrons in a np covalent bond are in high free energy state and electrons in a polar covalent bond are in low free energy state.
2. Changing bonds from np covalent to polar covalent is exergonic
3. Going from high free energy of np bonds to low free energy of polar bonds releases all the energy for cellular respiration.
What role(s) does NAD+ and FAD play in many reactions? What is the reduced form of both?
NAD+ and FAD are general electron acceptors in many reactions; and both are vitamin derivative coenzymes. The reduced form of NAD+ is NADH with a high free energy state.
FADH2 is the reduced form of FAD.
Define Allosteric enzyme regulation. What can it do and what can they be?
Allosteric enzyme regulation is a broad category of reversible enzyme regulation, including non-competitive inhibition.
It can active or inactive an enzyme.
The regulator can be either a small molecule activator or a protein activator.
Key: regulator binds away from the active site, changing enzyme conformation.
(Worth 1000 pts.)
What are the two important functions of polysaccharides?
What are the fuel storages in plants and animals? Where are they located?
List and define(where) examples of the 2nd function of polysaccharides.
Fuel storage-sugar and Structural Integrity
Starch: polysaccharide storage form of alpha-glucose in plants and Glycogen: polysaccharide storage form of alpha-glucose in animals.
Starch is located in chloroplasts, Glycogen located in mitochondria.
(all are beta glucose)Structural integrity in:
Cellulose: structurally rigid component of plant cell walls
Chitin: component of insect exoskeletons and fungal cell walls
Peptidoglycan: component of bacterial cell walls.
What is the reaction of ATP? What part of the reaction releases energy, and which needs energy? In other words what happens to it?
ATP's phosphate bonds are hydrolyzed(releases energy) and regenerated in metabolism(requires energy).
Slide 37.
What is Chemiosmosis?
Mechanism for oxidative phosphorylation and for Photophosphorylation
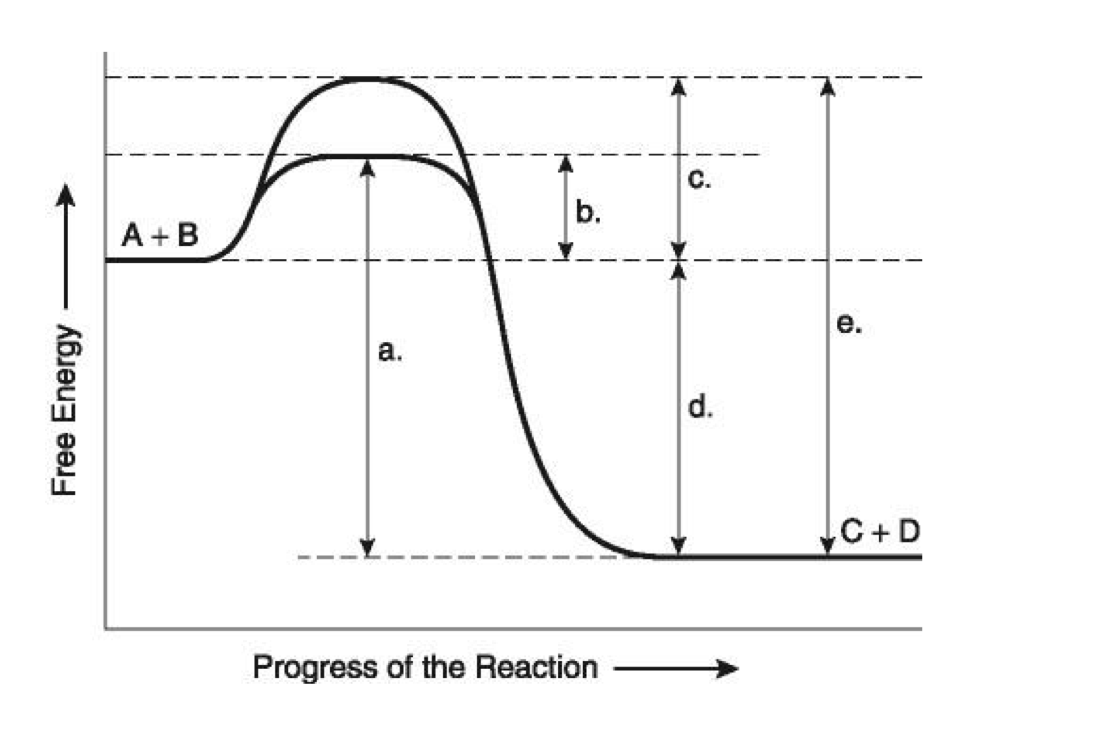 What do arrows a-e represent on the diagram?
What do arrows a-e represent on the diagram?
b: The activation energy of the reaction in the presence of an enzyme.
c. The activation energy of the reaction in the absence of an enzyme .
d. The delta G of the reaction
e. The activation energy it takes in the absence of an enzyme for the reverse/ backwards reaction