Elements of Group 1A are called?
Alkali Metals
Who is smaller?
Lithium or Fluorine
Fluorine
Who is Larger
Chlorine or Sodium
Chlorine`
What is Electronegativity?
A measure of the tendency of an atom to attract a bonding pair of electrons.
Which atom has a larger radius?
Cl-1 ion
Cl
Cl-1
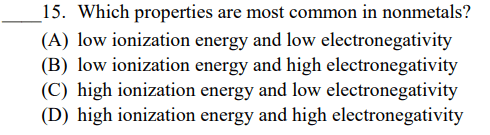
D

Cynthia Erivo

Coldplay

Clemson
What MLB exhibition season takes place in Florida and Arizona before Opening Day?
Spring Training
How many Groups are there in the Periodic Table?
18
Elements of Group 2A are called?
Alkaline Earth Metals
Who is larger
Lithium or Francium
Francium
Who has a smaller Ionization Energy?
Francium or Lithium
Francium
Who has a larger electronegativity?
Oxygen or Nitrogen
Oxygen
What happens to the size of an atom when it becomes a cation? Why?
Cations are atoms that have LOST electrons to become POSITIVELY charged. This causes an increase in the proton-electron ratio, increasing the coulombic attraction.

D

Ariana Grande

The Killers

University of Miami
What annual vacation period for many college students happens in March?
Spring Break
Elements of Group 7A are called?
Halogens
Magnesium or Calcium
Magnesium
What is Ionization Energy?
Energy required to remove an electron, and become an ion
Who has a larger electronegativity?
Bromine or Iodine
Bromine
Arrange each group of elements in order of increasing ionization energy.
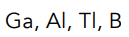


C

Halle Berry
Everytime
Lucky
Overprotected
Oops I Did It Again
If You Seek Amy
Britney Spears

University of Arizona
What famous rock musician known as “The Boss”
Bruce Springsteen
Elements of Group 8A are called?
Noble Gases
Who is Larger
Scandium or Chromium
Scandium
Who has a smaller Ionization Energy?
Barium or Magnesium
Barium
Metals have a ____________ electronegativity, while non-metals have a _____________ electronegativity.
Weak
Strong

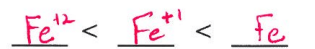

A
Conan O'Brien


University of Stanford
What city in Massachusetts is home to the Basketball Hall of Fame?
Springfield
Which scientist came up with the "Law of Octaves"
John Newlands
Elements of Group 6A are called?
Chalcogens
Who is Larger?
Iodine or Chlorine
Iodine
Which atom has a larger Ionization Energy?
1s22s22p63s1 or 1s22s22p63s2
1s22s22p63s2
Noble Gases have an electronegativity value of ___________
0
What does the Electron Affinity refer to?
Electron affinity is defined as the change in energy (in kJ/mole) of a neutral atom (in the gaseous phase) when an electron is added to the atom to form a negative ion.

Selena Gomez
Benny Blanco

Elton John

Syracuse
What last name belongs to the controversial daytime talk show host who hosted a chaotic tabloid-style show from 1991–2018?
(Jerry Springer)
