Lesson 16 - Comic Strip
Lesson 17 - Electronegativity
Lesson 18 - Polarity
Definitions
Fun Facts about Mrs. Valenton
100
If the polar bears represent atoms and the ice cream scoops represent electrons, what type of bond is represented in the illustration below?
What is non-polar covalent?
100
If the electronegativity difference between two atoms is greater than 2.4, what type of bond will likely form?
What is Ionic Bond
100
Draw the partial positive and partial negative charges for NH3


What is 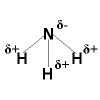

100
Define Electronegativity.
What is Describes the attraction of an atom toward electrons.
100
What is her favorite color?
What is purple.
200
In a polar covalent bond, how are the electrons share?
What is shared unequally?
200
Which type of element does not have an electronegativity value?
What is Noble Gases?
200
Draw the dipole for NH3
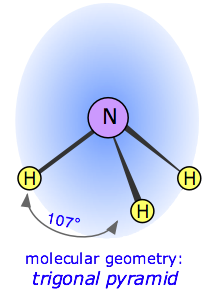

What is see board?
200
Define polarity.
What is Differences in electronegativity between atoms.
200
How old is her baby?
What is 5 months.
300
Explain why this picture shows a polar covalent bond for HCl.
What is there is an unequally sharing of the electrons where the electrons spend more time around the more electronegative atom
300
What type of bond forms between two atoms if the difference between their electronegativities is 0.38?
What is Non Polar Covalent?
300
Calculate the EN difference for the NF molecule.
What is 0.94?
300
Define a polar covalent bond.
What is The bonded electrons spend more time around the more electronegative atom and there are differences in electronegativity.
300
How long has she taught at JSerra?
What is 2 years?
400
What does each ice cream scoop represent?
What is one electron.
400
Based on its position on the periodic table, name an element that has an electronegativity value less than Lithium?
What is Look at group one.
400
What is the EN difference between SO2? What type of molecule is this?
What is 0.86 and polar?
400
Define a non polar covalent bond.
What is The bonded electrons are equally shared between the atoms and they have same electronegativities.
400
Where did she go on her honeymoon?
What is Spain and Portugal?
500
Explain what is happening between the polar bear and penguin and how it affects the electrons?
What is the polar bear is the more electronegative atom and it completely takes the electrons away from the penguin, which is the less electronegative atom
500
Where are the atoms with the greatest electronegativity values located?
What is Upper right corner of the periodic table?
500
Draw the dipole for aluminum trichloride and state the type of bond it forms.
What is see board?
500
What is the big difference between an ionic bond and a polar bond?
What is In an ionic bond, the electron is given up to the more electronegative atom. In a polar bond, the electron spends more time around the more electronegative atom.
500
How did her and her husband meet?
What is through a mutual friend at a birthday party.