What is the Kinetic theory?
What is all matter consist of tiny particles that are in constant motion?
What does inter-molecular attractions do in liquids?
What is reduce the amount of space between the particles in a liquid?
Describe the particles in a solid?
What is tightly packed and close together?
What is called when a substance changes from solid to vapor without passing the liquid sate?
What is sublimation?
Define Kinetic Energy
What is the energy an object has because of its motion?
Describe the motion of gas particles
What is rapid, constant, and random?
What is used to measure the vapor pressure of a liquid?
What is a manometer?
How many crystal systems are there?
What is seven systems?
Fill in the blanks
Phase diagrams shows the relationship between ________, __________, ____________.
What is temperature, pressure, and the phases of matter?
Define a Vacuum
What is an empty space with no particles and no pressure?
what is the SI unit of pressure
what is Pascal (Pa)?
Why does liquids evaporate faster when heated?
What is the average kinetic energy of its particles increases?
What happens to the particles of a solid when heated?
What is the particles vibrate more rapidly as their kinetic energy increases?
Fill in the blank
Organic chemists use sublimation to _______ mixtures and to ________ compounds.
What is separate?
What is purify?
Define an Amorphous solid
What is a solid that lacks an internal structure?
Fill in the blanks
One standard atmosphere is the pressure required to support ______ mm of mercury barometer at ____ degrees Celsius.
What is 760 mm of mercury ?
What is 25 ° Celsius?
Fill in the blank
a dynamic equilibrium exists between the vapor and the liquid because the rate of __________ of liquid equals the rate of ____________ of vapor.
Evaporation
Condensation
What is the difference between a crystal lattice and a unit cell?
What is crystal lattice refers to the structure of a crystal. The unit cell is a repeating part of the crystal structure?
What is the triple point temperature and pressure for water?
What is temperature is 0.016° C?
What is pressure is 0.61 kPa?
Define a Barometer
What is a device the is used to measure atmospheric pressure
Fill in the blanks
The _______ between particles are perfectly elastic so that the total _________ remains constant.
What is collisions?
What is Kinetic energy?
Florida is 105 m above sea level, water boils at 100 degrees Celsius. What happens to the Boiling point of water from Fl to Denver (1600 m above sea level).
What is the boiling point decreases?
Name 3 crystal systems
What is Cubic, Tetragonal, Othorhombic, Monoclinic, Triclinic, Hexagonal, and Rhombohedral?
Explain what a curve line on a phase diagram represent.
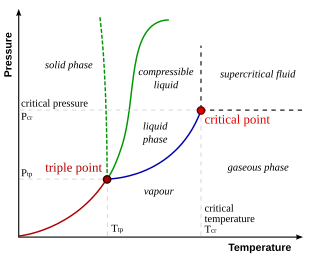
What is it represents two phases that exist in equilibrium?
Define Allotropes
What is 2 or more different molecular forms of the same element in the same physical state?