Anything that has volume and mass is called a(n)….
A. Element B. Matter C. Atom D. Proton
B: Matter
What subatomic particle has an overall negative electric charge and is found outside the nucleus of an atom?
A. Electron
B. Proton
C. Neutron
D. Acid
A: Electron
A substance that is composed of atoms of two or more different elements that are chemically combined is called a(n)…….
A. Mixture B. Solution C. Compound D. Acid
C: Compound
True or False: Gases have no definite shape and no definite volume.
True
A combination of two or more components that retain their identities is called a(n)……
A. Mixture B. Base C. Solution D. Compound
A: Mixture
What two things must you add together to get an elements mass number?
A. Protons and Electrons B. Electrons and Neutrons
C. Protons and Neutrons D. Neutrons and Ions
C: Protons and Neutrons
True or False: The nucleus of an atom has an overall positive electric charge.
True
True or False: Table salt (NaCl) is an example of a solution.
False (NaCl is a compound)
Process by which a cooling gas changes into a liquid and releases thermal energy.
A. Evaporation B. Freezing
C. Sublimation D. Condensation
D: Condensation
A solution containing a substance that produces hydrogen ions (H+) in water is called a(n)…..
A. Compound B. Acid C. Base D. Mixture
B: Acid
What are the two most abundant elements in the universe?
A. Oxygen and Nitrogen B. Hydrogen and Oxygen
C. Carbon and Hydrogen D. Hydrogen and Helium
D: Hydrogen and Helium
A substance that cannot be broken down into simpler substances by physical or chemical means is called a(n)….
A. Atom B. Nucleus C. Matter D. Element
D: Element
A mixture such as beach sand or oil and water in which the components are not uniformly distributed throughout its volume and can easily be seen separately is called a(n)
A. Solution B. Homogeneous Mixture
C. Heterogeneous Mixture D. Acidic Mixture
C: Heterogeneous Mixture
Process by which a solid slowly changes into a gas without first entering the liquid state.
A. Condensation B. Sublimation
C. Deposition D. Evaporation
B: Sublimation
A solution with a pH value of 10 is a……
(A). Acid (B). Base (C.) Neutral
B: Base
All elements on the periodic table are classified and arranged according to each elements……
A. Physical Properties B. Amount of matter
C. Chemical Properties D. Atomic Mass
C: Chemical Properties
What two subatomic particles are found in the nucleus of atoms?
1 correct = 400 points
2 correct = 800 points
1. Protons
2. Neutrons
An atom of the element Iron contains 26 protons, 30 neutrons, and 26 electrons. What is the atomic number and mass number of Iron?
1 correct = 400 points
2 correct = 800 points
Atomic Number = 26 (Number of Protons)
Mass Number = 56 (Protons + Neutrons)
Melting, freezing, evaporation, condensation, sublimation, and deposition are all processes that causes changes in the state of matter. Which three of these processes need the release of thermal energy to occur?
1 correct = 400 points
2 correct = 800 points
3 correct = 1,200 points
1. Freezing
2. Condensation
3. Deposition
Which is the more acidic solution: A solution with a pH value of 5 or a solution with a pH value of 2?
The solution with a pH value of 2 is more acidic.
What are the four states of matter?
1 correct = 500 points
2 correct = 1,000 points
3 correct = 1,500 points
4 correct = 2,000 points
1. Solid
2. Liquid
3. Gas
4. Plasma
What are the three subatomic particles found in all atoms?
1 correct = 500 points
2 correct = 1,000 points
3 correct = 1,500 points
1. Protons
2. Neutrons
3. Electrons
An atom of the element silver contains 47 protons, 61 neutrons, and 47 electrons. What is the atomic number and mass number of silver?
1 correct = 500 points
2 correct = 1,000 points
Atomic Number: 47 (Number of Protons)
Mass Number: 108 (protons + Neutrons)
Melting, freezing, evaporation, condensation, sublimation, and deposition are all processes that causes changes in the state of matter. Which three of these processes need the absorption of thermal energy to occur?
1 correct = 500 points
2 correct = 1,000 points
3 correct = 1,500 points
1. Melting
2. Evaporation
3. Sublimation
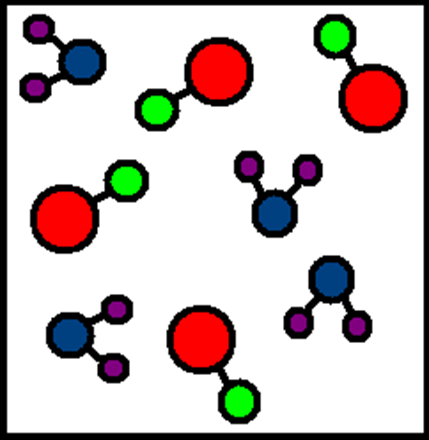
Mixture
What is the state of matter that occurs when collisions between particles are so violent that electrons are knocked away from atoms creating hot, highly ionized electrically conducting gases?
Plasma