What are the 3 types of mixtures?
Solutions, colloids, suspensions
Solution, colloid, or suspension: Mud
Suspension
What does molarity measure?
Concentration (mol/L)
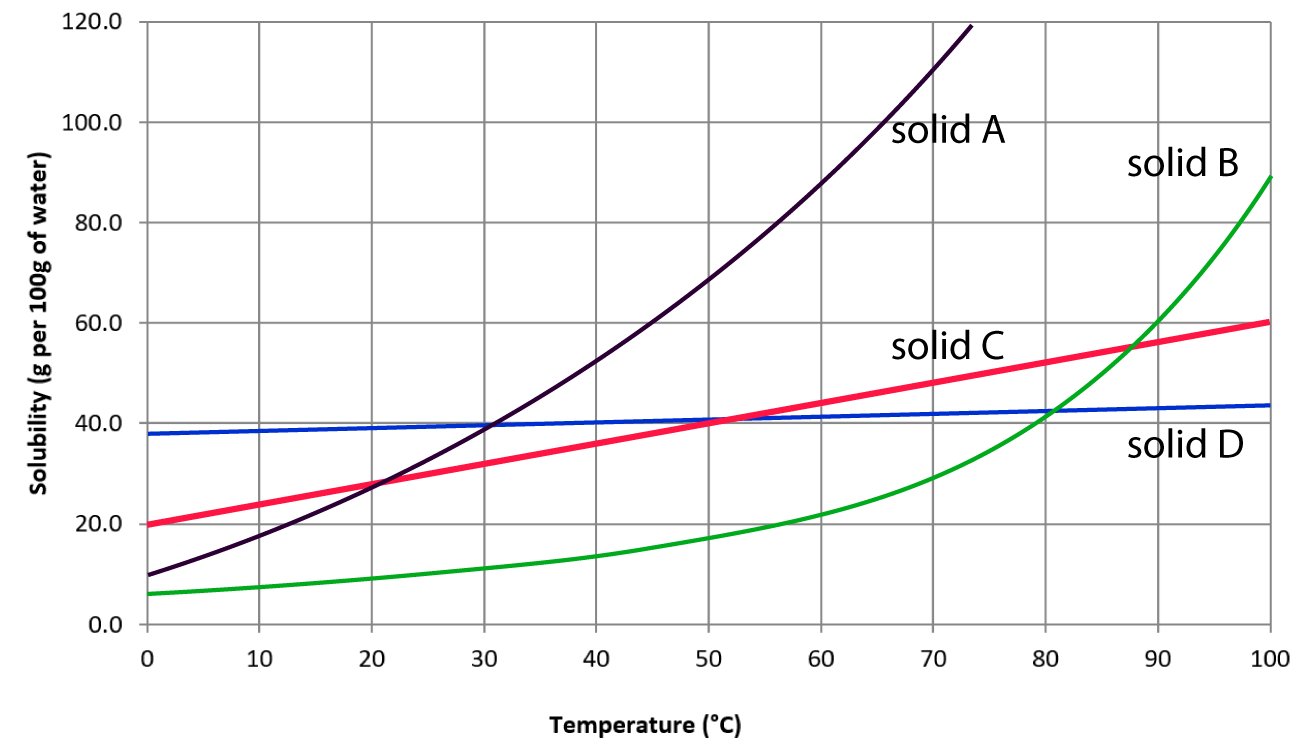
Which solid's solubility changes the LEAST?
Solid D
Increases or decreases: Increasing temperature __________ the solubility of solid solutes.
Increases
Rank the types of mixtures in order from smallest to largest particles.
Solutions, colloids, suspensions
Solution, colloid, or suspension: lemonade with pulp
Suspension
What is the equation for molarity?
M= mol/L

An aqueous solution of solid B at 30oC contains 40.0g of solid B. The solution is:
Supersaturated
Increases or decreases: Breaking up sugar cubes into powder __________ the solubility of the sugar.
Increases
What type of mixture has the largest particles, separates upon standing, can be filtered, and has the Tyndall effect?
Suspensions
Solution, colloid, or suspension: Air
Solution
What is the molarity of a solution containing 0.25 moles of sodium hydroxide dissolved in 3.5 liters of solution?
0.71 M

What is the solubility of solid A at 10°C?
About 18g
Increases or decreases: Soda making the "pssst" noise when opened. (Decreasing pressure ___________ the solubility of gases.
Decreases
What type of mixture has the Tyndall effect but does not separate?
Colloids
Solution, colloid, or suspension: Mayo
Colloid
How many liters of a 5 M solution can be made from 7.3 moles of NH3?
1.46 L

What temperature would a saturated solution containing 40 g of solid B need?
80oC
What's the name of substances that conduct electricity when dissolved (Ex: ionic compounds, acids, & bases)
Electrolytes
What type of mixture has the smallest particles and does not have the Tyndall effect?
Solutions
Solution, colloid, or suspension: bronze
Solution
How many moles of a 1.7 M solution can be made from 2.8 L of H2O?
4.76 mol

A student adds 80g of solid A at 30oC. What amount will not dissolve?
80g - 40g= 40g
What ions form when Ba(OH)2 dissociate when dissolved?
Ba+ and 2 OH-