When someone else does your experiment the same way
Replication
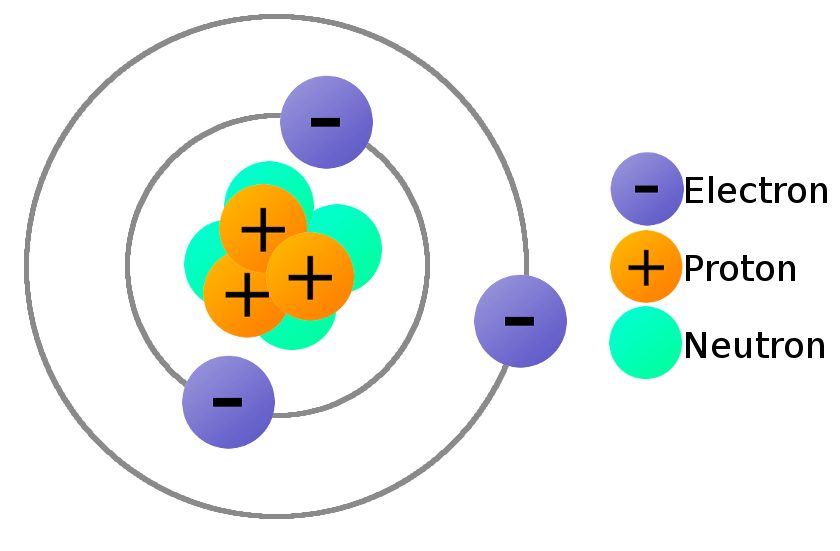
Draw an atom with 3 protons, neutrons, and electrons
Scratching a rock, smashing a rock, and putting an acid on it. Which a physical change?
Scratching and smashing
Lemon juice has a pH of 3. Is it an acid or base?
Acid
When an acid breaks something down what kind of change is it?
Chemical Change
How many independent variables should you have in an experiment?
One
Elements in the same ____________ share similar properties.
Same group/column
The amount of matter in an object
Mass

Jar of water and dirt is what kind of mixture?
Heterogenous Mixture
Tom checking his results by doing his experiment again.
Repetition
Theory or Law: New species came along because of adaptations. Why?
Theory - Explains

How many electrons and protons?
56
What is the volume of a rock if when placed in 20 mL of water, the water level rises to 300 mL?
280 mL
How are Pure Substances combined?
Chemically
What Geo means in Geocentric
Earth
Theory or Law: Mass is never created or destroyed, only transformed. Why?
Law. Describes
What region of the periodic table holds the bad conductor elements?
Nonmetals
The total mass of paper is 10g. What would be the total mass after it is burned halfway?
10 g
What kind of change is nuclear fusion and why?
Chemical because a new substance is made, light is given off, energy is released
State of matter where sound travels the fastest.
Solid
If people sleep more then they will be able to whistle longer. What is the independent and dependent variable?
IV: Amount people sleep
DV: Length of whistle
Name the subatomic particles, their charges, and their location in the atom.
Proton - Positive, Nucleus
Neutron - Neutral, Nucleus
Electron - Negative, Electron Cloud
A neutralization reaction is what kind of change and why?
Chemical. Salt is a new substance formed
A substance that looks the same throughout but can easily be separated. What is that called and give an example.
Homogenous Mixture. Example: Soda, air, salt water, etc
Depends on mass and gravity
Weight