How do particles move around in a gas?
Randomly
What will happen to the pressure in a system if the temperature in the system is INCREASED?
The pressure will increase.
What state of matter is represented by this particle diagram?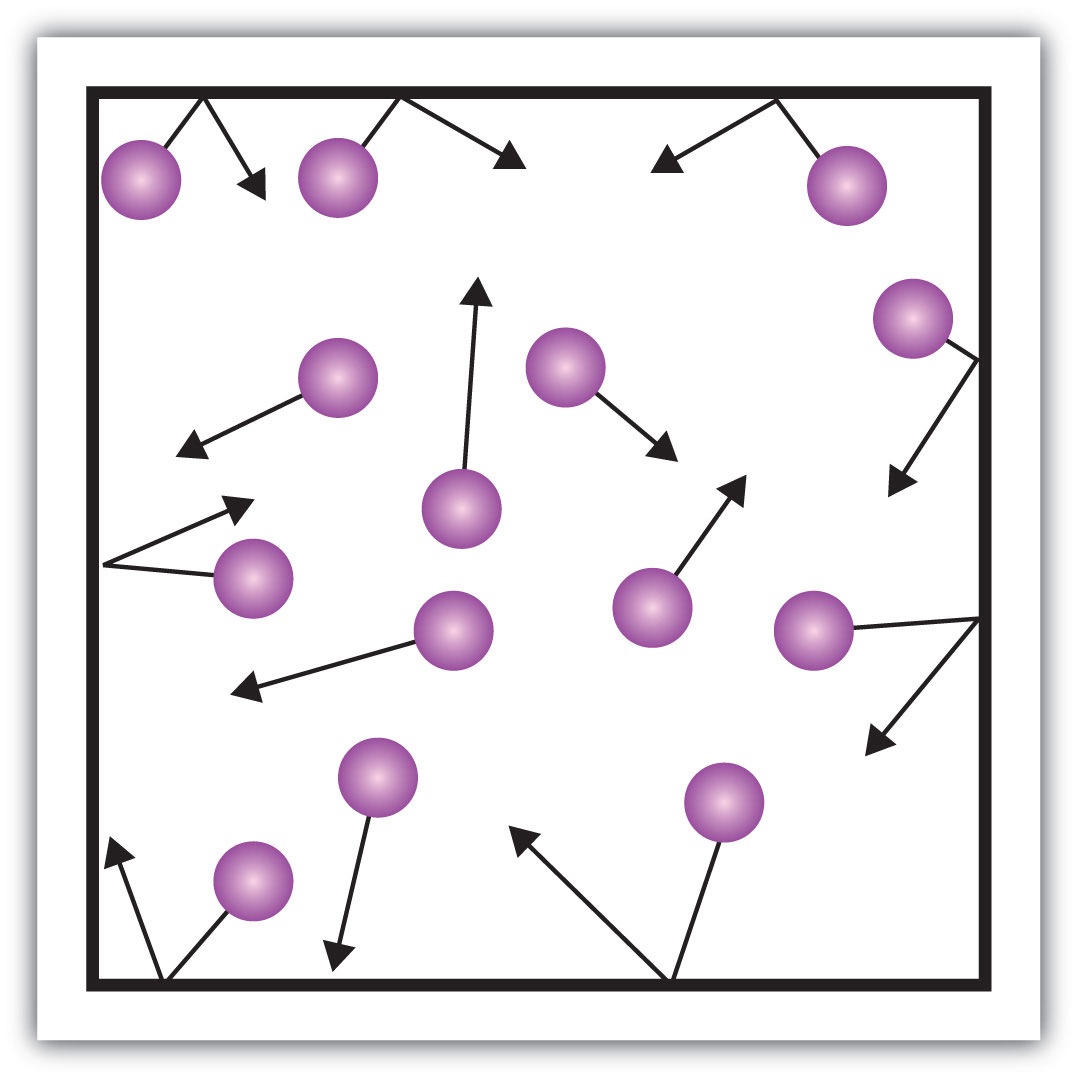
Gas
True or False: Vacuums add particles to a system
FALSE
According to the image, what is increasing as volume increases?
Temperature
What is the keyword for when particles move on the spot?
Vibrate
How are the number of particles and pressure related?
With more particles, collisions happen more often, which leads to a greater the pressure.
Draw a particle diagram of a liquid turning into a gas.
-->
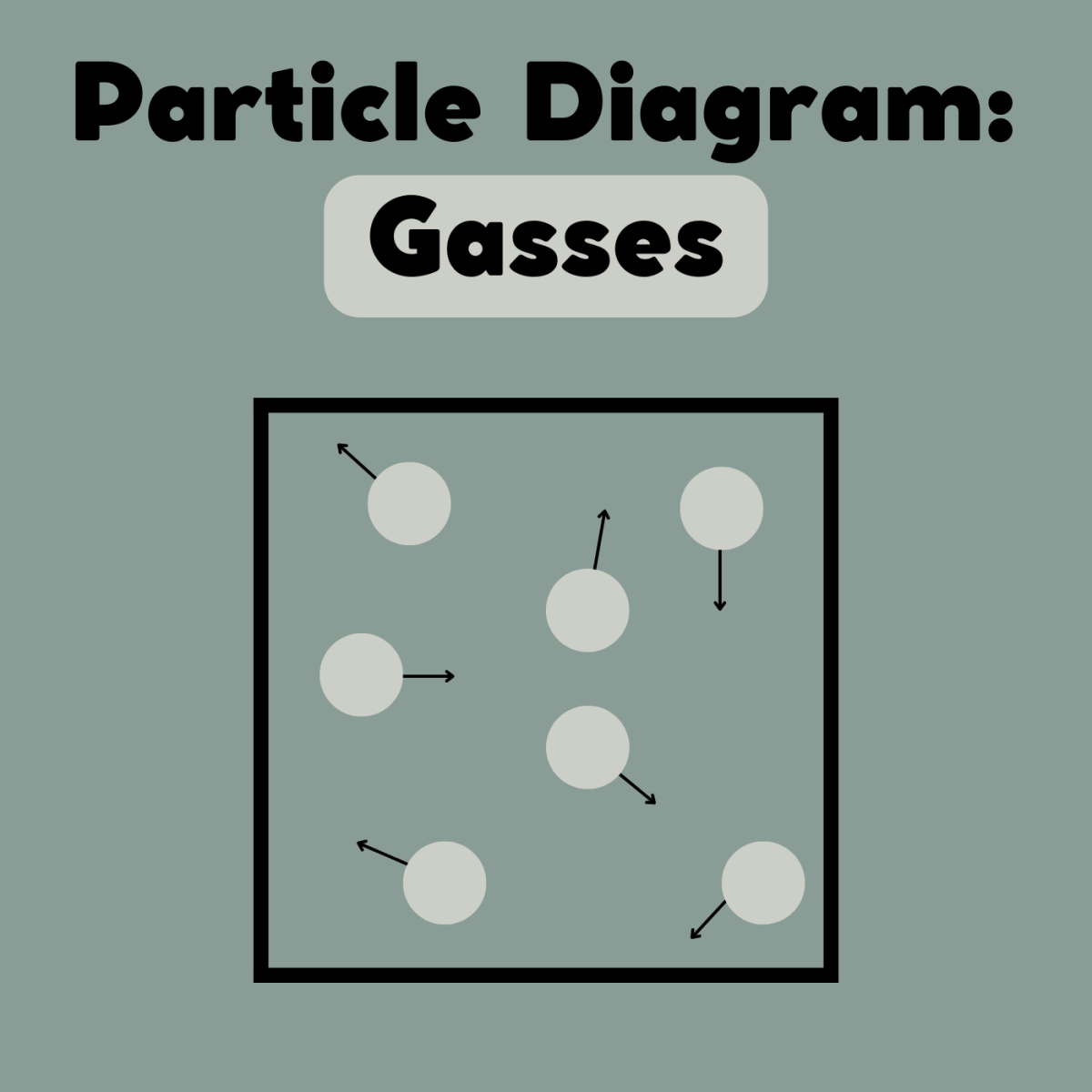
What does a vacuum do?
Removes particles from a system
Why can gases expand to fill their containers?
Gas particles move freely and are far apart, filling any available space.
More particles cause the pressure to...
Increase
If I INCREASE the volume in a system, what will happen to the pressure?
The pressure will decrease.
How could we represent QUICKER moving particles in a particle diagram? (Hint: think about the arrows)
Longer arrows/ lengthen arrows
What happened to the marshmallow when placed in the vacuum chamber?
The marshmallow expanded
Which phase of matter has a definite volume and an indefinite shape?
Liquid
What happens to particle speed and pressure when the temperature increases in a sealed container?
The particle speed increases and the pressure increases.
What are all the ways pressure can decrease.
By increasing volume, removing particles, and decreasing temperature.
Use the image to describe what is happening with regards to pressure.
As the volume in the container decreases, the pressure increases.
Why did the balloon expand in the vacuum chamber?
The air around the balloon was sucked out, which allowed the particles in the balloon to expand
Standing on your bed or lying down on your bed - Which will have a higher pressure? Explain why.
Standing - because it is a smaller surface area (your weight is not distributed)
What happens to air particles in a car tire and why does the car tire decrease air pressure during the winter? Explain your answer.
The cold causes air particles to decrease in speed causing less collisions which then causes less pressure.
Define pressure using the word “collisions.”
More collisions means higher force against the walls of the container, which means higher pressure.
Draw a particle diagram that ACCURATELY depicts what adding heat to a system would do to the particles.
*Shows something with a before having slower particles and after having faster moving particles*
Why did the vacuum make the water in the beaker boil?
Without air, there were no particles pushing on the surface of the water so it began to boil.
How could I represent an increase in pressure in this diagram?
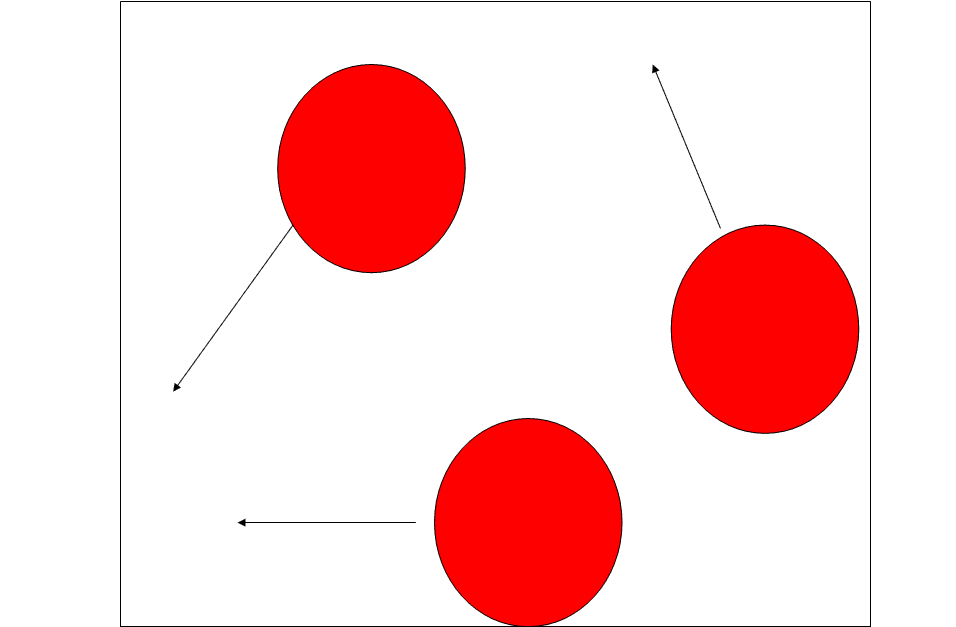
Either:
Add More particles
Faster Moving (Longer Arrow)
Flames Under System (Increased Heat)
Smaller Volume