Indicate which element in the pair has the greater atomic radius; sodium or lithium.
What is sodium?
Which element has a greater ionization energy; neon or boron?
What is neon?
This type of ion wants to gain electrons.
What is an anion?
The most electronegative element.
What is F?
Describe the trend of Atomic Radius down a group and across a period?
What is Atomic Radius increases down a group and decreases across a period?
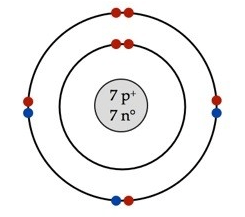 Name of ION and how did it become an ion..
Name of ION and how did it become an ion..
What is nitride by gaining 3 valence electrons?
Indicate which element has the greater atomic radius; strontium or magnesium.
What is strontium?
When ionizing, the level and sublevel that will lose electrons first in Cobalt.
What is 4s?
Calcium ion valence electrons do this and are classified as this type of ion.
What is lose 2 electrons and is a cation?
The most reactive non-metals.
What are halogens?
Describe the trends in Ionic Radius down a group and across a period.
What is the Ionic Radius increases down a group and decreases across a period for both metals and nonmetals?
Indicate which element has the greater atomic radius; carbon or germanium.
What is germanium?
Which element has the greater electronegativity: cesium, aluminum or Helium?
What is aluminum?
The oxidation state for Titanium (IV)
What is 4+?
Types of elements can change the amount of electrons lost when ionized.
What are transition metals?
The "father" of the the periodic table developed using properties and atomic masses.
Who is Mendeleev?
Which element has the greater atomic radius; selenium or oxygen.
What is selenium?
Arrange the following elements in order of increasing electronegativity; beryllium, magnesium, and strontium.
What is strontium, magnesium, and beryllium?
All metals create this type of ion.
What is a cation?
The elements that are inert.
What are noble gases?
He is credited for developing the Modern Periodic table using atomic numbers and properties.
Who is Moseley?
Explain why fluorine has a smaller atomic radius than chlorine.
What is because fluorine has fewer electrons?
How much energy it takes to remove the first electron from an atom.
What is ionization energy?
Phosphorus name when it gains 2 electrons in the outer shell.
What is phosphide?
Group of Elements with similar properties to the image.
with similar properties to the image.
What are halogens?
List the non-metal elements with similar properties as the picture.

oxygen, sulfur, selenium