All matter is made up of atoms. What is the structure of an atom, the subatomic particles that make it up and their charges?
Protons (+) & Neutrons (0) in nucleus
Electrons (-) in electron cloud surrounding the nucleus
Occurs between 2 nonmetals on the periodic table.
(2 possible answers, name both for double the points!)
1.) covalent bond
2.) share electrons
Explain the difference between distance and displacement.
Distance is a scalar quantity that refers to how much ground an object has covered during its motion.
Displacement is a vector quantity that refers to the object's overall change in position.
Solve for Distance & Displacement.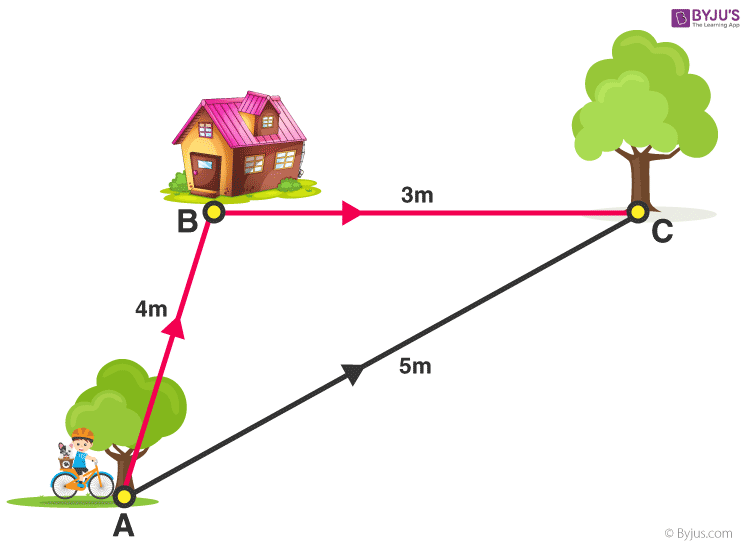
Distance = 7 m
Displacement = 5 m NE
Mg(OH)2
Name & Type of Bond
Magnesium Hydroxide
Ionic Bond
Matter is classified as ___________ or ____________ depending on its atomic makeup.
Pure Substances or Mixtures
Pentanitrogen Monoxide
Formula & Type of Bond
N5O
Covalent

A person travels from point A to B, then C before stopping. Solve for distance and displacement.
Distance = 15 m
Displacement = 11.2 m SE
A cat runs up a tree that is 10m high with a velocity of 2.2 m/s.
How long did it take the cat to reach the top?
*Must show all work (with proper units) on white board!*
4.5 seconds
Produces more Hydronium ions
Acids
Is it possible for a chemical and physical change to occur at the same time? Explain your answer and support it with an example.
Yes, burning something! Chemically becoming a new substance and physically changing the way it looks.
Other examples: Cooking, burning a candle & the wax melting.
List 3 differences between an Acid and a Base.
Acids: Sour taste, turn litmus paper red, create hydronium in water
Bases: Bitter taste, turn litmus paper blue, creates hydroxide in water
What occurred between 3.5s - 20s?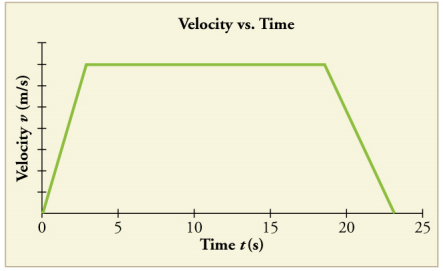
The object was at a constant velocity, in other words it was not accelerating.
Determine the mass of a basketball player who is running across the court with a velocity of 2.3 m/s with a momentum of 201.25 kg*m/s.
Show all work with proper units on the white board!
87.5 kg
This states that that any particle of matter in the universe attracts any other with a force.
This force depends on the product of the masses, and the distance between them.
Newton's Law of Universal Gravitation
Identify the element below, and explain what this diagram is called and what it is showing.
Atom of Sodium (Na)
Bohrs Model (shows all subatomic particles)
11 p+ & 12 n0 in the nucleus
11 e- in electron cloud
Balance & identify type of reaction
_ Pb(OH)2 + _ HCl -> _ H2O + _ PbCl2
1, 2, 2, 1
Double Replacement
A force of 100N was necessary to lift a rock. A total of 150 joules of work was done. How far was the rock lifted?
*Must show all work and units on the white board!
W = F x d
d = W/F
d = 150 J / 100 N
d = 1.5 meters
Explain what is happening to the PE and KE of the ball as it moves from position A to G.
Hint: think of a roller coaster!

*Why is this possible? (for 100 extra points!)
Position A has MAX PE and no KE.
As it moves to position C the PE is decreasing and KE is increasing.
At D the ball will have its maximum velocity and KE.
As D moves to G, KE will decrease will moving uphill, converting back to PE.
*This is possible because of the Law of Conservation of Energy (energy cannot be created or destroyed, only transformed!)
List Newton's 3 Laws (names & descriptions) and give 1 example to support each one.
1.) Law of Inertia: Object at rest stays at rest, and an object in motion stays in motion unless acted on by an unbalanced force.
2.) F = ma: Force exerted by an object is equaled to the product of its mass and acceleration.
3.) Action-Reaction: For every action there is an opposite and equal reaction.
*Examples may vary!*
All isotopes of technetium are radioactive, but they have widely varying half-lives. If an 800.0 g sample of technetium-99 decays to 100.0 g of technetium-99 in 639,000 years, what is its half-life?
*show all work & units on the white board!
213,000 years
_ AlBr3 + _ K2SO4 ->_ KBr + _ Al2(SO4)3
Balance & identify any cations/anions
(must answer all to earn any points!)
2, 3, 6, 1
All compounds present are cations!
Name and describe the 2 types of nuclear processes considered for energy production.
Give 1 example of each.
Explain the pros and cons for each process.
What 2 things do they have in common?
Fission (splitting of a radioactive atom, usually with Uranium rods in power plants) Fusion (combination of 2 hydrogen atoms to form a heavier helium atom, occurs on the sun and other stars)
Fission is currently used in power plants (nuclear weapons in the past) because it is much easier to control with the technology we have. One downside is the long half lives from the radioactive material.
Fusion creates a tremendous amount of energy, but we do not have the technology to sustain such high temperatures needed for the reaction (could be devastating if something went wrong.)
Both are forms of nuclear energy and CLEAN (no pollutants!!!)
A 3rd grader is playing soccer at recess. The kid has a mass of 23 kg, velocity of .5 m/s and is on a hill that is 5 meters in height.
Identify the type of energy and solve for it.
*Must show all work and units on the white board!
Energy Type: Kinetic
KE = (1/2)(m)(v2)
KE = (1/2)(23)(.52m/s)
KE = (11.5 kg)(0.25 m/s)
KE = 2.875 Joules
Explain the importance of physical science.
Hint: be specific!
Physical Science is all science that is not life science.
Physical science explains much of what you observe and do in your daily life. In fact, you depend on physical science for almost everything that makes modern life possible!
Ex: You couldn’t drive a car, create a text message, or sky dive without decades of advances in chemistry and physics. You wouldn’t even be able to turn on a light!
TLDR: Physical science explains everyday observations and actions. Its advances make modern life possible!