This happens to an electron during the ionic bonding process.
What is transfer from metal to nonmetal?
This happens to electron(s) during the covalent bonding process.
What is electron(s) are shared?
The classification of the reaction:
4Al + 3O2 --> 2Al2O3
What is synthesis?
The taste of an acid.
What is sour?
The pH scale is based on the concentration of this ion.
What is hydronium?
The prefix for seven.
What is hepta?
A metal typically become this in an ionic bond.
What is a cation or positive ion?
The compound listed that is covalent: NO2, NaCl, CaCl2, or Li2O.
What is NO2?
The classification of the reaction:
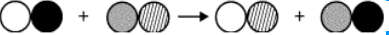
What is double replacement?
The type of solution that affects litmus by turning blue and phenolphthalein by turning pink.
What is a base?
A solution has a pH of 7 which would be considered this type of solution.
What is neutral (or water)?
The correct name for Al2S3.
What is Aluminum Sulfide?
A property of an ionic compound when it is dissolved in water.
What is conduct electricity or become an electrolyte?
A description of the melting point of a covalent compound compared to the melting point of an ionic compound.
What is lower?
The coefficient that would be placed in front of the Al in the reaction:
Al + O2 --> Al2O3
What is 4?
How Bronsted-Lowry described a base.
What is a proton acceptor?
A substance that is reading a pH of 13 would be considered this type of base.
What is strong?
The correct name for N2O3.
What is dinitrogen trioxide?
The reason that ionic bonds do not form a true molecule.
What is the ions are attracted by their positive or negative charges OR the ions do not remain joined together?
The polarity of the covalent compound H2O.
What is polar?
The complete balanced equation for the reaction:
AgNO3 + NaCl --> AgCl + NaNO3
What is AgNO3 + NaCl --> AgCl + NaNO3? (already balanced)
An acid is defined by the amount of this produced when dissolved in water.
What is hydronium?
The weakest pH of an acidic solution.
What is 6?
What is Li3P?
The Lewis Dot structure for an ionic bond between Sodium and Oxygen
..
What is 2[Na]+1 + [:O:]-2?
..
What is a pair of electrons?
The mass of the products of a candle that has burned inside a closed container for 4 minutes (the beginning mass of the candle is 500 g).
What is 500 g?
An acid where very little of the hydronium has been ionized would be considered this type of acid.
What is weak?
What is 7?
The correct formula between atoms of Magnesium and Iodine.
What is MgI2?