On the Periodic Table, the Atomic number of an element tells you how many of which positive particle is in the nucleus of an atom?
Protons
When thermal energy moves between two objects at different temperatures and must be touching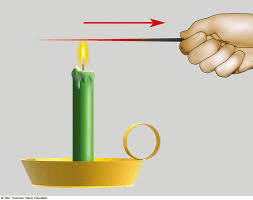
Conduction
An unknown solution has a pH less than 6. Which of the following would describe the solution?
(base, acid, neutral)
acid (anything below 7 is acidic)

Motion that occurs after 30 seconds
Slowing down, decelerating, negative acceleration
Which property of the Sun's radiation do scientists use to classify the EMS radiation into different categories?
Wavelength

What subatomic particle do the dots on the orbital rings outside the nucleus represent?
Electron
Point where KE is highest and lowest
Kinetic Energy highest - Point X
Kinetic Energy lowest - Point W
The temperature at which an element or compound changes from solid to liquid.
Melting point
The motion happening from 5:00 to 5:15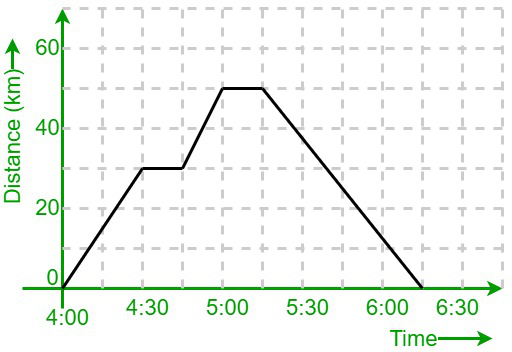
At rest, no motion
Type of light wave behavior seen here.
Refraction
Based on the arrangement of Periodic Table, elements are organized in vertical columns called ___________.
Elements in the same column share similar __________.
Group/Family, Properties
John puts his spoon into a bowl of fresh, hot mashed potatoes as shown in the figure below.
Which direction does energy flow when the two objects come together?
(Thermal energy is transferred in the form of heat from the __________ to the __________)
Thermal energy is transferred in the form of heat from the warmer mashed potatoes to the cooler spoon.
The substances that are combined to start a chemical reaction are called _________
The new substances formed in a chemical reaction are ________
Reactants, Products
The speed of two different objects is shown on the graph below. Describe the speed of the object represented by the blue line, compared to the red object.
The blue line object moves at a slower speed than the red line object.
When NASA scientists communicate with the Mars rover, they use electromagnetic radiation with a long wavelength and low energy.
Which type of EMS radiation do they most likely use?
Radio waves
When 2 or more different atoms bond together, they create a _________, forming a new substance.
Compound
What type of energy transformation is occurring at the coal power plant?
__________ energy is transformed into _________ energy.
Kinetic/Mechanical, Electrical
Mass per unit volume of an object as compared to pure water.
(How many particles in a certain volume of space?)
Density
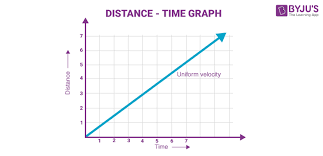
Type of motion that this graph indicates
Constant motion/speed
(as time increases, distance increases)
Describes the electromagnetic spectrum:
Longer wavelengths have _____ frequencies.
Shorter wavelengths have _____ frequencies.
Low
High
Number of protons, electrons and neutrons in Potassium

19 - protons
19 - electrons
20 - neutrons
Based on the information in the table, what can be inferred about the kinetic energy of the particles in the air as the temperature increases throughout the day?
The kinetic energy of particles in the air increases as the temperature increases during the day
A student grew crystals on a string by adding salt to a container of water, hanging a string in the solution, and allowing the water to evaporate. They took four containers, added the same amount of water, a different amount of salt, and identical lengths of string to each one. After two weeks, the student removed the strings and crystals, and weighed them to determine the mass of the crystals grown in each solution.
What is the test variable (independent variable) in this experiment?
amount of salt
What would be the magnitude and direction of the net force acting on the car in the diagram?
75 N Leftward
Remote controls and thermal imaging technology use invisible light with a lower frequency than the visible range. What is this part of the electromagnetic spectrum called?
Infrared