A substance that is only made up of one type of atom is a called a(n) ______________.
Element
Soil is an example of a ______________ mixture.
heterogenous
What is the equation for density?
D = m/v
Particles in a solid.....
A: Do not move
B: Move freely
C: Vibrate
D:Slide past one another
C: Vibrate
Give an example of a physical change that has not been mentioned yet.
Tons of options bruh
Which of the following is an mixture?
A: Soil
B: Oxygen (O2)
C: Carbon Dioxide (CO2)
D: Pure Water
A: Soil
Salt water is an example of a __________ mixture.
homogenous
If I am measure the amount of mL of a liquid I am measuring __________.
Volume ONLY
 What are A, C, and D?
What are A, C, and D?
B: Liquid
C: Gas
Give an example of a chemical change that has not been mentioned yet.
67
Which of the following is an compound?
A: Soil
B: Oxygen (O2)
C: Carbon Dioxide (CO2)
D: Salt Water
C: Carbon Dioxide (CO2)
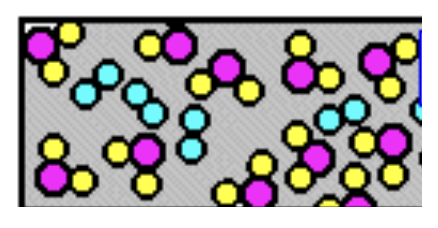 What is this a mixture of?
What is this a mixture of?
Mixture of elements and compounds
3A 6 gram block has a volume of 2 cm3. What is it's density?
3 g/cm3
________ is the property that describes how well a substance can be bent/hammered without breaking.
Malleability
Which of the following is an example of a chemical change?
A: Breaking down carbon dioxide into carbon and oxygen
B: Melting ice
C: Water condensing on a cold glass
D: Tearing a piece of paper in half
A: Breaking down carbon dioxide into carbon and oxygen
Which of the following is a pure substance?
A: Homogenous mixtures
B: Heterogenous mixtures
C: Compounds
D: Solutions
C: Compounds
Give an example of a heterogenous mixture AND a homogenous mixture that has not been previously mentioned.
Hmmmmm interesting answer......
Substances that are more dense will _________.
Substances that are less dense will __________.
Sink, Float
Give an example of a substance with high viscosity
Honey or Maple syrup are the go to's......
What are the THREE ways in which an object can physically change?
Change in size, shape, phase/state of matter
Give an example of an element, a compound and a mixture that has not been previously mentioned.
Do you need to study more?
Give an example of a heterogenous mixture AND a homogenous mixture that has not been previously mentioned.
Good job!....maybe.....
What is the mass of a cube that has a density of 5 g/mL and has a volume of 10 mL?
50 grams

What are B and D (both ways) AND why is the line flat at those points?
B Left = Freezing B Right = Melting
C Left = Condensation C Right = Evaporating
Flat line means temp does not change during phase change
What are the FIVE ways in which a substance can chemically change?
Gas produced
Precipitate
Color change