Diamond, graphite, and graphene look and behave very differently. Why are all three still classified as the same element?
They are made of the same type of atom (carbon) arranged in different structures.
Which statement correctly describes a compound?
A. It contains only one type of atom
B. It contains atoms that are not bonded
C. It contains two or more kinds of atoms chemically bonded together
D. It can be separated by physical means
C. It contains two or more kinds of atoms chemically bonded together
How are particles distributed in a heterogeneous mixture?
A. Evenly
B. Unevenly
B. Unevenly
How are particles distributed in a homogeneous mixture?
A. Evenly
B. Unevenly
A.evenly
Why do elements in the same group have similar chemical properties?
A. They have the same atomic mass
B. They have the same number of protons
C. They have the same number of valence electrons
D. They are all metals or all nonmetals
C. They have the same number of valence electrons
Elements in the same group on the periodic table have similar:
(chemical) properties
A compound differs from a mixture in that a compound always has a
a. Homogeneous composition
B. Heterogenous composition
c. Minimum of three components
D. maximum of three components
A.
True or false: heterogenous mixtures can be a mixture of solids, liquids, and/or gases.
True.
Mixtures can be any combination of all states of matter: solids, liquids, and gases.
You observe that a sample of clear liquid has evaporated but left a powder like solid residue in the bottom of the container. how this liquid would most likely be classified.
a solution / homogenous mixture
Why do elements on the right side of the periodic table tend to gain electrons?
A. They have large atomic radii
B. They have low electronegativity
C. They are trying to fill their valence shell
D. They have many energy levels
C. They are trying to fill their valence shell
Which statement best explains why atomic models have changed over time?
A. Atoms physically changed as technology improved
B. New evidence and experiments led scientists to revise models
C. Scientific laws replaced atomic theories
D. Scientists stopped agreeing on atomic structure
B. New evidence and experiments led scientists to revise models
Rust forms when iron reacts with oxygen to form iron oxide. Explain why rust is considered a compound.
Rust is a compound because iron and oxygen are chemically bonded to form iron oxide.
A student observes a sample containing oil droplets floating in water. Even after stirring, the droplets remain visible. How should this sample be classified?
A. Element
B. Compound
C. Homogeneous mixture
D. Heterogeneous mixture
Heterogeneous mixture. Since, Oil and water do not mix evenly and form visible layers or droplets, which is a defining characteristic of a heterogeneous mixture.
Explain why air is considered a homogeneous mixture even though it is made of different gases.
Air is homogeneous because its gases are evenly distributed and cannot be seen separately. Every sample of air has the same composition, which makes it uniform throughout.
If an element is shiny, conducts electricity, and loses electrons easily, where is it most likely located?
A. Right side of the periodic table
B. Bottom right corner
C. Left side of the periodic table
D. Group 18
C. Left side of the periodic table
Why do scientists continue to use atomic models even though atoms cannot be seen directly?
A. Atomic models show the exact size of atoms
B. Atomic models replace laboratory experiments
C. Atomic models help visualize the structure of atoms
D. Atomic models prove that atoms exist
C. Atomic models help visualize the structure of atoms
Which picture represents a pure substance?
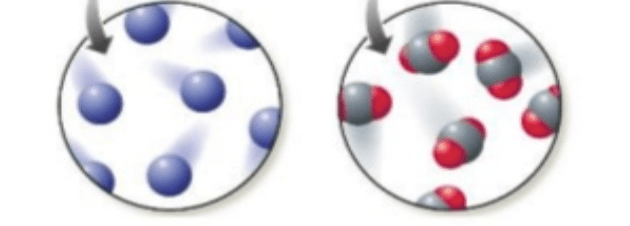
Both
A student claims that a mixture is homogeneous because it looks uniform at first glance. Explain why this claim may be incorrect and describe how time or physical processes could reveal whether the mixture is actually heterogeneous.
A mixture may look uniform at first but still be heterogeneous. Over time, heavier particles may settle or liquids may separate into layers. Stirring, filtering, or letting the mixture stand can reveal uneven distribution of substances, showing that the mixture is heterogeneous.
A clear liquid contains two substances but cannot be separated by filtration. What mixture is this and why?
Homogeneous mixture. Filtration separates visible solid particles from liquids. If the substances cannot be separated by filtration, it suggests the particles are very small and evenly distributed, which is characteristic of a homogeneous mixture.
Sodium reacts violently with water, but sodium chloride (table salt) is safe to eat. Explain how periodic trends and chemical bonding help explain this difference.
Sodium is highly reactive on its own because it easily loses one electron. When sodium bonds with chlorine, it forms sodium chloride, a stable compound. Chemical bonding changes the properties of the elements, making salt safe to eat.
Which element is least likely to react with others?
A. Fluorine
B. Sodium
C. Neon
D. Oxygen
C. Neon
Which of the following substances is classified as a compound?
A. Oxygen gas
B. Carbon dioxide
C. Air
D. Iron
B. Carbon dioxide
What defines a mixture?
A. A mixture can be broken down into its basic substances through physical means.
B. A mixture cannot be broken down into its basic substances through physical means.
C. A mixture is created through a chemical reaction
D. A mixture is described by a chemical formula
A. A mixture can be broken down into its basic substances through physical means.
What is another term we can use for homogeneous mixtures? (Hint it contains a solute and solvent.)
Solution
Why is hydrogen placed at the top of Group 1 even though it is not a metal?
Hydrogen has one valence electron like Group 1 elements. It is placed there because of its electron structure, even though its properties are different.
Which category best describes elements found on the left side of the periodic table?
A. Nonmetals
B. Metalloids
C. Noble gases
D. Metals
D. Metals
State any two differences between a compound and a mixture
compounds can not be separated by physical means while mixtures can
Compounds are pure substances, mixtures are not
Fresh orange juice with seeds is often labeled as “natural” and “pure.” Explain why this juice is scientifically classified as a heterogeneous mixture rather than a pure substance.
Fresh orange juice with seeds is a heterogeneous mixture because the seeds do not dissolve in orange juice. The seeds and juice remain physically separate and unevenly distributed, even though the juice may look uniform.
Why are separate particles not visible in a homogeneous solution?
A. One substance dissolves in the other
B. The particles settle at the bottom
A. One substance dissolves in the other
Why are noble gases often used in light bulbs and signs?
Noble gases do not react easily, so they are safe to use. Some noble gases also glow when electricity passes through them. For ex: neon