Draw Lewis Structure for HCN?

Draw the electron configuration for O.
1s2 2s2 2p4 or He 2s2 2p4
Balance the equation:
KMnO4 + HCl = KCl + MnCl2 + H2O + Cl2
2 KMnO4 + 16 HCl = 2 KCl + 2 MnCl2 + 8 H2O + 5 Cl2
C3H8 + O2 -------> CO2 + H2O
a) If you start with 34.8 g of C3H8 and 5.2 g of O2, determine the limiting reagent
O2
This movie involved a robot trash compactor showing a post-apocalyptic earth
Wall-E
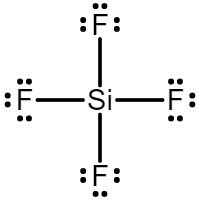
Draw the Lewis Structure for SiF4
Draw the electron configuration for Cl.
What was the First Pixar Movie
Toy Story
Convert 500g of CH4 to moles
31.17 Moles of Propane
Draw the electron configuration for Kr.
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6
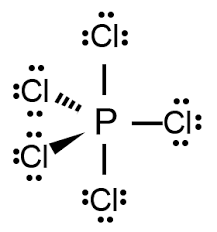
Draw Lewis Structure PCl5
This movie had a main character obsessed with finding a twinkie
Zombieland
Calculate the frequency of radiation with a wavelength of 442 nm.
(1m = 109nm) (C=2.997*108m/s) (λν=C)
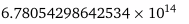 1/s
1/s
A 40.0 g sample of ethanol releases 2952 J as it cools from 50.0 °C. Calculate the final temperature of the ethanol q = mcΔT
q = mcΔT -2952 J = (40.0 g)(2.46 J/g C/C)()T) -2952 J = (98.4 J//C)()T) )T = - 30 /C
This Sports Icon was also known as the great Bambino, the Sultan of Swat.
Babe Ruth
Draw the Lewis Structure H2SO4.

Draw the electron configuration for I.
 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p5
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p5
Name the movie where the quote "I have a big head and little arms. I'm just not sure how well this plan was thought through."
Meet the Robinsons
The Kardashian sisters' father, Robert Kardashian, is known for defending which famous person in α 1994 murder trial.
O.J. Simpson
How many moles of HCL are in a 500L HCL solution with a Molarity of 20M
Who was the first woman to win a Nobel Prize?
Marie Curie
Draw the electron configuration Br
[Ar] 4s² 3d¹⁰ 4p⁵
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p5
Please give me the Molecular Geometry for SiO2.
Linear
A student must use 225 mL of hot water in a lab procedure. Calculate the amount of heat required to raise the temperature of 225 mL of water from 20.0 °C to 100.0 °C. the density of water is 1.00 g /mL
q = mcΔT
Since the density of water is 1.00 g /mL, the mass of 225 mL of water is 225 g - c for water is 4.184 J/g C°C q = mc)T q = (225 g)(4.184 J/gC°C)(100.0°C - 20.0 °C) = 755312 J = 75.5 kJ
Determine the empirical formulas for compounds with the following percent compositions:
15.8% carbon and 84.2% sulfur
CS2