True or False: An ionic bond is usually between a nonmetal and a nonmetal.
False. An ionic bond is usually between a metal and a nonmetal or a polyatomic ion. A covalent bond is usually between a nonmetal and a nonmetal.
How many atoms of oxygen are in Al2(CO3)3?
9 atoms of oxygen
How many atoms are in 4.5 moles of C?
2.7 x 1024 atoms C
How many moles of oxygen are necessary to react completely with 3.4 moles of propane (C3H8)?
C3H8 + 5 O2 --> 3 CO2 + 4 H2O
17 mol O2
tin (IV) oxide
What is the name of the compound SO2?
Sulfur dioxide
What are the reactants in the following chemical equation?
CH4 + O2 --> H2O + CO2
CH4 + O2
What is the molar mass of LiNO3?
68.944 g/mol LiNO3
What mass of N2 is produced from the decomposition of 102 g NaN3?
2 NaN3 --> 2 Na + 3 N2
65.9 g N2
Consider a single bond, a double bond, and a triple bond. List the bond types in order of decreasing bond strength.
triple, double, single
What is the chemical formula for copper (II) sulfate?
CuSO4
What type of reaction is the following reaction?
CaCO3 --> CaO + CO2
decomposition
How many formula units are in 89.3 g MgSO4?
4.47 x 1023 formula units MgSO4
50.0 g of Mg and 75.0 g of HCl react to form MgCl2 and H2. What is the limiting reactant?
Mg + 2 HCl --> MgCl2 + H2
HCl is the limiting reactant
An electronegativity difference equaling 0 would result in what type of covalent bond forming between two atoms?
nonpolar covalent bond
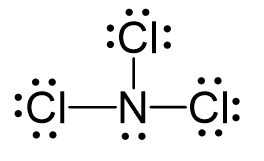
Draw the Lewis Dot Structure for NCl3
Balance the following skeleton equation:
FeS + O2 —> Fe2O3 + SO2
4 FeS + 7 O2 —> 2 Fe2O3 + 4 SO2
What is the molarity concentration of a solution containing 3.22 moles of NaCl in 0.520 L of water?
6.19 M NaCl
Determine the theoretical yield, in grams, of Al2O3 when 43.1 g of Al reacts with O2.
4 Al + 3 O2 --> 2 Al2O3
81.4 g Al2O3
What is the molecular geometry of a molecule made up of a total of 3 atoms that has 2 lone pairs of electrons and 2 shared pairs of electrons?
bent
What is the molecular geometry of a molecule with 1 lone pair of electrons and 3 bonds?
trigonal pyramidal
Predict the products of the following reaction:
AlF3 + I2 -->
No Reaction!
How much water must be added to 1.50 L of a 1.8 M CaCl2 solution to make a 0.10 M solution of CaCl2?
27 L
Calculate the percent yield of S2Cl2 if the theoretical yield is 98.3 g S2Cl2 and a student recovered 96.4 g S2Cl2 in the lab.
98.1% S2Cl2
How many moles of MgCO3 are required to form 3.00 L of 1.75 M MgCO3 solution?
5.25 mol MgCO3