Which of the following quantitative measures of inspiratory and expiratory flow is especially useful in diagnosing chronic obstructive pulmonary disease (COPD)?
A. Forced expiratory volume in one second (FEV1)
B. Forced vital capacity (FVC)
C. Functional residual capacity (FRC)
D. Peak expiratory flow (PEF)
A. Forced expiratory volume in one second (FEV1).
This, along with FVC (choice B), helps distinguish obstructive from restrictive lung disorders. Choice C: FRC does not measure airflow and is therefore incorrect. Choice D: PEF is used primarily to monitor asthma patients.
A 27-year-old woman is diagnosed with acute femoral vein thrombosis. She has systemic lupus erythematosus diagnosed 3 months ago. Her only medication is hydroxychloroquine.
On physical examination, temperature is 37.9 °C (100.2 °F); other vital signs are normal. A faint malar rash is noted. The left leg shows 2+ edema.
Activated partial thromboplastin time (aPTT) 39 s
Hemoglobin 10.8 g/dL (108 g/L)
Leukocyte count 3200/μL (3.2 × 109/L)
Platelet count 302,000/μL (302 × 109/L)
Prothrombin time 11s
A mixing study does not correct the aPTT. Lupus anticoagulant is positive, and high-titer IgG β2-glycoprotein and IgG cardiolipin antibodies are measured.
Which of the following is the most appropriate immediate treatment?
A. Apixaban
B. Dabigatran
C. Fondaparinux
D. Low-molecular-weight heparin
The most appropriate treatment for this patient is initial low-molecular-weight heparin (LMWH) and transitioning later to warfarin (Option D).
She has systemic lupus erythematosus and now has a likely diagnosis of antiphospholipid antibody syndrome (APLAS). APLAS is an autoimmune disorder in which thrombosis and fetal demise (in pregnancy) may occur. Patients with APLAS are at risk for arterial and venous thrombosis. The diagnosis is based on the clinical criteria of thromboembolism or pregnancy morbidity and laboratory findings of medium or high-titer antiphospholipid antibodies present on two or more occasions at least 12 weeks apart. A clue to the presence of the lupus anticoagulant is activated partial thromboplastin time prolongation that fails to correct with a mixing study. This patient's diagnosis of APLAS is based on the presence of an acute thrombotic event and positive test results for all three antiphospholipid antibodies (lupus anticoagulant, anticardiolipin antibody, and β2-glycoprotein antibody). This patient's antiphospholipid measurements should be repeated after 12 weeks for final confirmation. Extended anticoagulation is needed now. The risk for recurrent thrombosis is reported to be higher in patients with “triple-positive” APLAS serologic tests when treated with a direct oral anticoagulant (DOAC) compared with LMWH followed by dose-adjusted warfarin.
In general, the preferred treatment of acute venous thromboembolism in patients with APLAS is LMWH followed by warfarin. DOACs may be associated with a higher risk of thrombosis compared with warfarin in patients with “triple-positive” APLAS or in patients with arterial thrombosis. Therefore, apixaban and dabigatran would not be the most appropriate choices for this patient (Options A, B).
Evidence supporting the use of parenteral anticoagulants other than LMWH is extremely sparse and consists primarily of a few case reports. Fondaparinux cannot be recommended as an alternative to LMWH until sufficient data can confirm its effectiveness and safety (Option C). Finally, fondaparinux has no reversal agent. Caution should be used in patients at risk for bleeding because the half-life is 17 hours.
A 49-year-old woman is evaluated in the emergency department for sudden-onset severe headache of 24 hours' duration. The headache reached its peak intensity within 1 minute. She is otherwise well and takes no medications. On physical examination, vital signs are normal. Nuchal rigidity is present. Findings on mental status and funduscopic examinations are normal. There are no cranial nerve deficits. A CT scan of the head is normal.
Which of the following is the most appropriate next step in management?
A. Cerebral catheter angiography
B. Lumbar puncture
C. Magnetic resonance venography of the brain
D. Oral sumatriptan
The most appropriate next step in management is lumbar puncture (Option B).
The patient is seen 24 hours after sudden onset of severe headache (thunderclap headache). Thunderclap headache is defined as a severe attack of headache pain developing abruptly and reaching maximum intensity within 1 minute. Although occasionally of primary origin, thunderclap headache is a medical emergency that warrants immediate diagnostic evaluation. Thunderclap headache has a broad differential diagnosis; the most serious is aneurysmal subarachnoid hemorrhage (SAH). A small proportion (less than 5%) of patients with aneurysmal SAH will have a CT scan of the head that is negative for bleeding and require a lumbar puncture to rule out this condition. In patients with SAH, results on lumbar puncture performed 12 to 24 hours after symptom onset will reveal elevated erythrocyte counts and xanthochromia (yellow color caused by breakdown of red blood cells). If lumbar puncture is performed within 12 hours of symptom onset, it may be challenging to distinguish a traumatic process from the acute presence of erythrocytes from aneurysmal bleeding because xanthochromia may not yet have developed.
Cerebral catheter angiography (Option A) is not appropriate because SAH has not been confirmed and an invasive diagnostic test is not yet indicated. If evidence suggests SAH, vascular imaging is indicated to first evaluate for an aneurysm; usually this is done with a less invasive test, such as CT angiography. Noninvasive vascular imaging will also allow for evaluation of other causes of thunderclap headache, such as reversible cerebral vasoconstriction syndrome (RCVS). RCVS manifests as multifocal distal arterial narrowing on imaging.
Magnetic resonance venography of the brain (Option C) is not indicated because the patient has no historical features or risk factors for dural sinus thrombosis, such as a hypercoagulable state or oral contraceptive use; SAH is the initial possible diagnosis of highest risk. If SAH is ruled out, venous imaging should be considered, particularly if CT findings suggest thrombosis (hyperdensity on noncontrast CT of the head).
Oral sumatriptan (Option D) should not be administered because the patient has an atypical presentation for migraine, and sumatriptan may worsen outcomes in patients with aneurysmal SAH or RCVS.
In 2025, this fluffy character from Pop Mart became a ubiquitous presence from handbags to parade floats.
Labubu
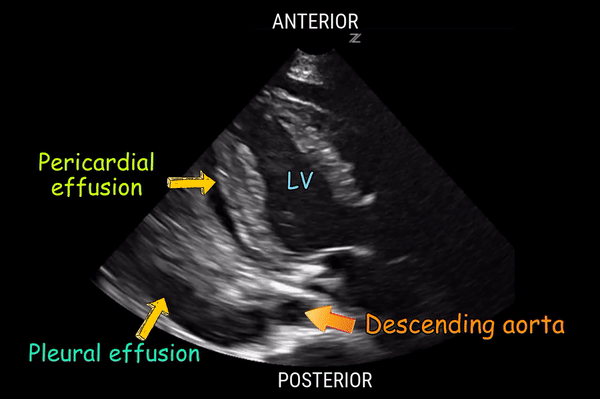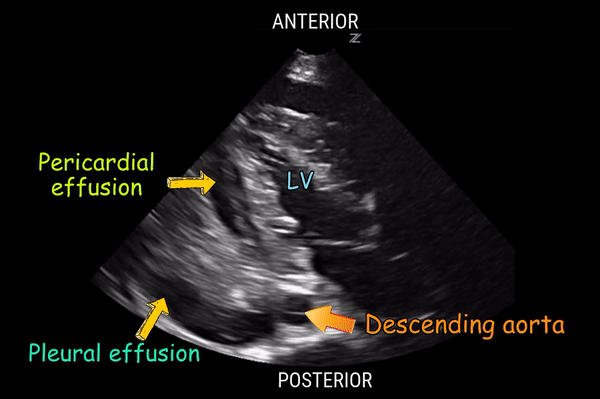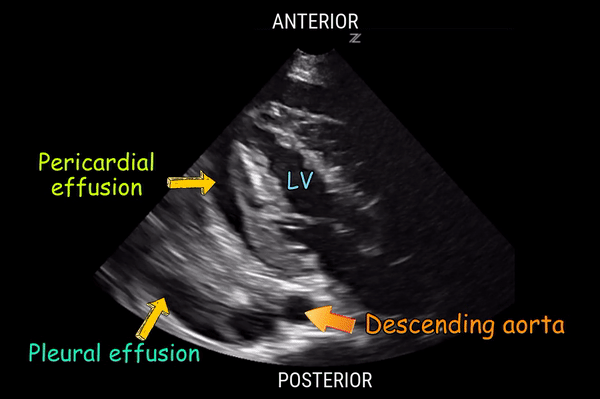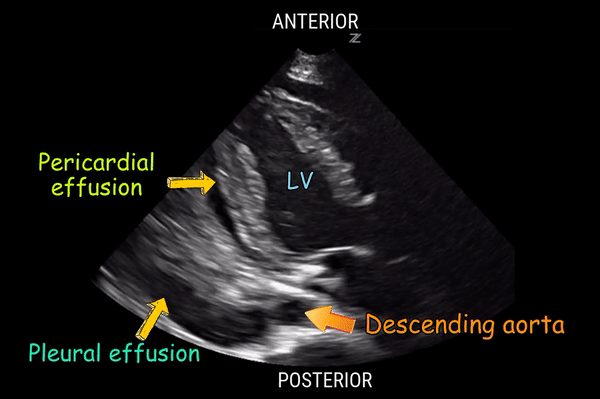
Anechoic fluid anterior to the descending aorta in the parasternal long-axis (PLAX) view is consistent with which of the following diagnoses?
A. Pleural effusion
B. Pulmonary edema
C. Ascites
D. Pericardial effusion
D. Pericardial effusion
Which of the following physiologic changes is most likely indicative of an obstructive pulmonary disorder?
A. FEV1 is decreased; FVC is decreased; TLC is decreased
B. FEV1 is decreased; FVC is decreased; TLC is increased
C. FEV1 is decreased; FVC is increased; TLC is increased
D. FEV1 is increased; FVC is decreased; TLC is increased
B. FEV1 is decreased; FVC is decreased; TLC is increased.
These readings, in addition to decreased FEV1/FVC and normal or increased residual volume, are indicative of an obstructive pulmonary disorder. Choice A is indicative of restrictive pulmonary disorders. Choice C is not indicative of a pulmonary disorder, and choice D is indicative of a mixed pulmonary disorder.
A 35-year-old man is evaluated in the emergency department for a 2-day history of swelling in his left leg. His mother was diagnosed with a venous thromboembolism at age 50 years. The patient can identify no provoking event. He is otherwise well and takes no medications. On physical examination, vital signs are normal. The entire left lower extremity is swollen. Lupus anticoagulant, anticardiolipin antibody, and anti–β2-glycoprotein antibody testing is normal. Duplex ultrasonography confirms deep venous thrombosis in the left femoral vein.
Which of the following should be measured now to inform immediate treatment decisions?
A. Antithrombin
B. Factor V Leiden
C. Proteins C and S
D. No additional testing
No hereditary thrombophilia testing performed now will inform immediate treatment decisions (Option D).
Although identification of the inherited thrombophilias has advanced the understanding of venous thromboembolism (VTE) pathophysiology, it has little influence on clinical management, which does not differ acutely based on the presence of an inherited thrombophilia. Management duration is typically determined by whether the VTE event was provoked by a reversible or self-limited insult. In this patient with an unprovoked deep venous thrombosis, guidelines suggest that extended anticoagulant therapy should be considered regardless of the presence or absence of an inherited thrombophilia. The choice of extended thromboprophylaxis should be informed by bleeding risk and reevaluated each year. The choice of anticoagulant may be influenced by the presence of antiphospholipid antibody syndrome, but this patient's antiphospholipid evaluation was unremarkable.
Antithrombin III (ATIII) is a natural anticoagulant that can lead to thrombophilia in the setting of an inherited or acquired deficiency. The prevalence of inherited ATIII deficiency is approximately 1 in 3000 to 5000 persons and leads to a strong predisposition for VTE. ATIII is reduced during an acute VTE, and measurement at this time would not provide useful information (Option A).
Factor V Leiden (FVL) is the most common inherited thrombophilia (Option B). Heterozygous FVL is most common in patients of European ancestry, affecting up to 5% of this population. FVL can be detected by genetic testing (DNA testing) or a functional coagulation test for activated protein C resistance. Although genetic testing now would not affect immediate treatment options for this patient, testing can be performed independent of the presence of an acute thrombosis. Therapy with a direct thrombin inhibitor (argatroban, dabigatran) or an oral factor Xa inhibitor (apixaban, edoxaban, rivaroxaban) can result in falsely normal activated protein C functional test results.
Proteins C and S are vitamin K–dependent proteins that degrade activated factors V and VIII (Option C). Inherited deficiencies are generally associated with thromboses before the age of 50 years and are associated with a strong family history of thrombosis. These proteins are also reduced in the setting of acute VTE, and measurement now would not provide useful information or influence immediate treatment.
A 44-year-old man is evaluated for a 15-year history of headaches that have become increasingly frequent over the past 6 months, now occurring between 15 and 20 days per month. He describes the headache pain as a global feeling of heaviness or tightness that seems to improve with exercise or distraction. Headaches are often preceded and accompanied by neck tightness. He has had no photophobia, phonophobia, nausea, aura, or neurologic symptoms. Acetaminophen, relaxation training, and cognitive behavioral therapy have been ineffective. NSAIDs caused gastroesophageal reflux symptoms. All physical examination findings, including vital signs, are normal.
Which of the following is the most appropriate preventive treatment?
A. Amitriptyline
B. Butalbital
C. Cyclobenzaprine
D. Onabotulinum toxin A
E. Topiramate
The patient has tension-type headache and should be treated with amitriptyline (Option A).
The most prevalent primary headache condition, tension-type headache is defined as a headache disorder that, unlike migraine, is mild to moderate in intensity and not associated with nausea or severe sensory or neurologic symptoms. Annual population prevalence in the United States is approximately 40% for episodic (<15 days monthly) tension-type headache and 2.5% for its chronic (15 or more days monthly) counterpart. Despite its high population prevalence, tension-type headache comprises only 3% of outpatient headache consultations, most likely because of its nondisabling nature. Pain is typically bilateral, nonpulsatile, and mild to moderate in intensity without aggravation by physical activity. Acetaminophen, aspirin, NSAIDs, and caffeine-containing compounds are typically effective acute treatments for tension-type headache, and many patients self-medicate successfully. Nonpharmacologic treatments have gained popularity as alternative interventions for tension-type headaches in recent years. Psychological treatment strategies, such as relaxation training and cognitive behavioral therapies, are evidence-based and should be considered for all patients with tension-type headache. Available options for prevention of tension-type headache are more limited. Amitriptyline is the only agent shown to be effective in controlled trials; venlafaxine and mirtazapine have more limited data.
Due to potential for habituation and medication overuse, guidelines recommend that butalbital (Option B) be avoided in the management of primary headache disorders, such as migraine and tension-type headache.
Many with tension-type headaches also will exhibit pericranial and cervical muscle tenderness on examination. This finding became the basis for the development of this specific headache diagnostic category. Despite this association, evidence suggests muscle relaxants such as cyclobenzaprine (Option C) are ineffective in this condition.
Onabotulinum toxin A (Option D) is indicated in the management of chronic migraine, but studies in tension-type headache have been negative.
Topiramate (Option E) is a first-line preventive treatment for episodic migraine (<15 days monthly) and has evidence of benefit in chronic migraine (15 or more days monthly). However, it has no evidence of benefit in tension-type headache.
At the start of the 2026 awards season, this annual Hollywood event awarded “KPop Demon Hunters” Best Animated Movie and featured anti-ICE pins worn by several celebrities.
Golden Globe Awards
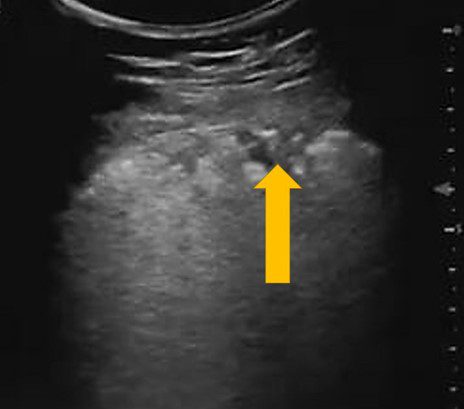
The image above was obtained in Zone 3 of a patient with confirmed a COVID-19 polymerase chain reaction test (PCR).
What lesion is the yellow arrow pointing to?
A. Pleural effusion
B. Pulmonary infarct
C. Subpleural consolidation
D. Air bronchograms
C. The arrow is pointing to a pleura based hypoechoic lesion. The findings are consistent with subpleural consolidation. No anechoic collection is seen.
A 29-year-old woman was hospitalized 24 hours ago with hypoxemic respiratory failure due to influenza pneumonia. She was intubated and placed on mechanical ventilation. Treatment includes lactated Ringer solution, intravenous peramivir, propofol, fentanyl, and norepinephrine. On physical examination, temperature is 38.1 °C (100.6 °F), blood pressure is 109/59 mm Hg, pulse rate is 90/min, and respiration rate is 24/min. The arterial PO2 is 60 mm Hg on FIO2 of 0.65, and positive end-expiratory pressure is 10 cm H2O. Tidal volume is 6 mL/kg ideal body weight, and plateau pressure is 27 cm H2O. The patient is sedated but wakes to touch and is calm. Pulmonary rhonchi are present bilaterally. Chest radiograph shows bilateral opacities. Echocardiogram reveals normal cardiac function and chamber size.
Which of the following is the most appropriate intervention to improve oxygenation?
A. Increase level of sedation
B. Perform recruitment maneuver
C. Prone positioning
D. Start diuretics
The most appropriate management is prone positioning (Option C).
The patient has severe acute respiratory distress syndrome (ARDS) due to influenza pneumonia (arterial PO2/FIO2 = 92). Prone positioning reduces compression of portions of the lung behind the cardiac and mediastinal structures and improves ventilation/perfusion matching in patients with ARDS. Prone positioning for at least 12 to 16 hours daily should be considered for patients with moderate to severe ARDS with persistent hypoxemia despite use of low tidal volume ventilation and plateau pressure less than 30 cm H2O. The indications for prone positioning have been variably defined, but entry criteria in a pivotal clinical trial that showed benefit included: arterial PO2/FIO2 <150; FIO2 ≥0.60 on positive end-expiratory pressure >5 cm H2O. Study participants were intubated for less than 48 hours before initiating prone positioning. Although the patient is also meeting goals for lung protective ventilation, randomized trials show that the addition of prone positioning improves mortality in those patients.
The sedation level should not be increased (Option A). The patient is meeting goals for sedation and is calm and easily arousable. Light-sedation protocols or daily awakening trials are associated with decreased length of mechanical ventilation and length of ICU stay.
In patients with refractory hypoxemia, a recruitment maneuver (Option B)—applying a high level of CPAP to open collapsed alveoli (e.g., continuous pressure to 35 cm H2O for 40 seconds)—has been conditionally recommended. However, a randomized control trial demonstrated that recruitment maneuvers in patients with moderate to severe ARDS was associated with increased risk for barotrauma, pneumothorax, and death compared with standard care.
The FACCT trial suggested that excessive fluid resuscitation is harmful to ARDS patients. This trial compared conservative with liberal fluid strategies based on central venous pressure and pulmonary artery occlusion pressure (a surrogate for left atrial pressure) in patients with ARDS. Although mortality did not differ between groups, patients who were treated with conservative fluid management showed improved oxygenation and decreased time on the ventilator and in the ICU. In patients who are hemodynamically stable and do not have end-organ hypoperfusion, minimizing fluid administration is warranted. This patient still requires norepinephrine blood pressure support; diuresis is not indicated at this time (Option D).
A 32-year-old man is evaluated in the emergency department for a 1-week history of fatigue, low-grade fever, bruising, and epistaxis. He has no other medical problems, and he takes no medications.
On physical examination, temperature is 37.8 °C (100.0 °F), blood pressure is 120/65 mm Hg, pulse rate is 108/min, and respiration rate is 22/min. Pallor is noted. Dried blood is present in the nares. Multiple bruises are seen on his extremities, and he has petechiae at his ankles.
Activated partial thromboplastin time 38 s
D-dimer 2.5 μg/mL (2.5 mg/L)
Hematocrit 22%
Leukocyte count 2300/μL (2.3 × 109/L)
Platelet count 22,000/μL (22 × 109/L)
Prothrombin time 25 s
Albumin 3.5 g/dL (35 g/L)
Alanine aminotransferase 30 U/L
Aspartate aminotransferase 35 U/L
Bilirubin, total 0.9 mg/dL (15.4 μmol/L)
Fibrinogen 60 mg/dL (0.6 g/L)
Which of the following is the most likely diagnosis?
A. Aplastic anemia
B. Coagulopathy of liver disease
C. Disseminated intravascular coagulation
D. Thrombotic thrombocytopenic purpura
The most likely diagnosis is disseminated intravascular coagulation (DIC) (Option C).
DIC results from the simultaneous activation of coagulation and fibrinolysis. It is associated with severe sepsis, usually with septic shock; with disseminated malignancy, most classically with mucin-secreting pancreatic adenocarcinoma; and in pregnancy, with complications of placental abruption and eclampsia. The initial pathogenesis involves widespread endothelial injury and circulating procoagulants that lead to disseminated microvascular thrombi, with consumption of platelets and clotting factors, and erythrocyte shearing injury leading to hemolysis. Fibrinolysis is accelerated, resulting in dissolution of the microvascular thrombus, usually before thrombotic complications are noted. Classic laboratory findings include thrombocytopenia, prolonged activated partial thromboplastin and prothrombin times (aPTT, PT), elevated INR, hypofibrinogenemia, and elevated D-dimer level. Management is directed primarily at the inciting cause of DIC and supported with platelet transfusions, cryoprecipitate, and fresh frozen plasma as needed. In this previously well, young patient with DIC associated with pancytopenia, an underlying leukemia should be suspected. A characteristic feature of acute promyelocytic leukemia is presentation with DIC at the time of diagnosis. Aplastic anemia can explain the patient's pancytopenia but does not account for other aspects of the coagulopathy that are present
A 43-year-old man is evaluated for a 6-year history of recurrent, severe migraine occurring every 3 months and lasting 2 to 3 days. He describes the migraine pain, which typically awakens him from sleep, as debilitating, intense, throbbing, and unilateral in location (although alternating sides). The migraine prohibits normal physical activity, such as going to work. For 30 to 45 minutes during a migraine episode, he may experience hemifield visual scotoma and ipsilateral numbness in the face and hand. Photophobia and nausea are intense, and he typically vomits within an hour of awakening on migraine days. Oral sumatriptan, oral rizatriptan, and nasal zolmitriptan have not relieved the pain. All physical examination findings, including vital signs, are normal.
Which of the following is the most appropriate next step in management?
A. Brain MRI
B. Head CT
C. Hydrocodone
D. Subcutaneous sumatriptan
The most appropriate step in management is subcutaneous sumatriptan (Option D).
The patient's migraine meets International Classification of Headache Disorders, 3rd edition, diagnostic criteria for migraine with aura. Aura may occur in 20% to 30% of patients with migraine. It frequently precedes pain but also occurs during or without head discomfort. Aura symptoms involve positive (e.g., paresthesia) and negative (e.g., scotomata) neurologic phenomena developing gradually and evolving over a period of 5 to 60 minutes. Resolution is gradual and complete. Typical aura involves any combination of homonymous visual, hemisensory, or language symptoms. This patient has a typical migraine-associated aura. The presence of typical aura does not affect choice of acute migraine therapy, although triptans are contraindicated in the setting of brainstem or hemiplegic auras. Given this patient's migraine with typical aura and the severe migraine present on awakening with rapidly developing vomiting, subcutaneous sumatriptan is an appropriate therapeutic choice.
Brain MRI (Option A) is the preferred brain imaging study in patients with chronic headaches and any red flags concerning for secondary headaches. This patient presents with a stable pattern of episodic headaches meeting criteria for migraine with aura. No red flags are present, and both the headaches and aura symptoms can alternate locations, reducing concerns for a focal lesion. In the presence of a stable clinical pattern of migraine and a normal neurologic examination, guidelines suggest that brain imaging is not indicated.
Head CT (Option B) is only indicated in evaluation of acute severe headache. Because this patient has a more episodic headache, head CT is not indicated.
Guidelines recommend against using opioid agents, such as hydrocodone (Option C) or butalbital products, in the management of primary headache disorders. Efficacy of hydrocodone in acute migraine has not been established. When compared with medications indicated in acute migraine (NSAIDs, triptans), opioid drugs carry increased risks of habituation, addiction, and development of medication overuse headache.
This A24 film stars Timothée Chalamet as a charismatic and obsessive table-tennis prodigy.
Marty Supreme
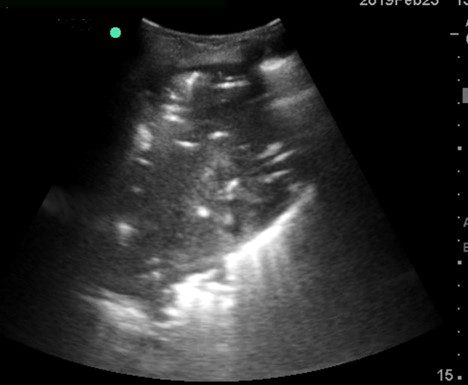
A 45-year-old female presented to the clinic with a history of fever, chest pain and dyspnea for 7 days. The physician performed a physical exam and then proceeded to perform a POCUS lung examination. The above view was obtained.
Which of the sonographic findings is seen in the image below?
A. Air bronchograms
B. Curtain sign
C. Subpleural consolidation
D. Spine sign
A. Air bronchograms
When the alveoli are filled with fluid/secretions, the affected lung area can be seen on ultrasound as the fluid/secretions allow the ultrasound beam to travel through it. If the bronchial tree still has air in it then we see echogenic areas within the lung parenchyma. These are known as sonographic air bronchograms. These could be tiny punctate lesions or could be longer and linear. If the air bronchogram is not moving during respiration it is known as a static air bronchogram. This indicates the presence of trapped air and is consistent with resorptive atelectasis. However, if the echogenic air bronchogram is moving with respiration, then it indicates the presence of a non-retractile consolidation and is known as a dynamic air bronchogram and is most probably due to a pneumonia (94% specificity).
A 36-year-old woman is evaluated for respiratory failure requiring invasive mechanical ventilation following a motor vehicle accident. On physical examination, temperature is 38.0 °C (100.4 °F), blood pressure is 100/65 mm Hg, pulse rate is 100/min, and respiration rate is 26/min. Oxygen saturation is 90% with an FIO2 of 0.50 and positive end-expiratory pressure of 12 cm H2O. There are multiple contusions on her chest. Estimated central venous pressure is normal. Other than tachycardia, cardiac examination is normal. There are diffuse lung crackles. Hemoglobin has remained stable. Echocardiogram reveals a normal ejection fraction and valvular function. ECG shows only sinus tachycardia.
ABG results are as follows:
pH 7.3
PCO2 50 mmHg
PO2 66 mmHg
Chest radiograph is shown below:

Which of the following is the most likely diagnosis?
A. Acute respiratory distress syndrome
B. Diffuse alveolar hemorrhage
C. Pulmonary edema
D. Pulmonary embolism
The most likely diagnosis is acute respiratory distress syndrome (ARDS) (Option A).
The diagnosis of ARDS is based on the 2012 Berlin criteria: onset within 1 week of known ARDS insult (most are within 72 hours); bilateral opacities on chest imaging consistent with pulmonary edema; respiratory failure not related to cardiac failure or volume overload; arterial Po2/FIO2 ratio less than 300 on at least 5 cm H2O positive end-expiratory pressure (PEEP). Once criteria are met, the severity of ARDS is based on the arterial PO2/FIO2 ratio: mild, >200 to <300; moderate, 100 to 200; severe, <100. This patient meets the diagnostic criteria for moderate ARDS. Her PO2/FIO2 ratio is 132. This patient likely has ARDS secondary to trauma. Diffuse alveolar hemorrhage (Option B) is the result of bleeding into the alveolar spaces secondary to the disruption of the alveolar-capillary basement membrane. Most patients present with hemoptysis, although this might be absent and the diagnosis nevertheless suggested by the appearance of new diffuse or focal infiltrates and falling hemoglobin level. Considering the relationship of the trauma to the onset of respiratory distress and absence of hemoptysis, ARDS is a more likely diagnosis.
Acute cardiogenic pulmonary edema (Option C) can present as hypoxemic respiratory failure. In patients who present with acute respiratory failure, it is important to evaluate for cardiogenic causes. Evaluation should include an assessment for volume overload (jugular venous distention, S3, peripheral edema), an ECG, echocardiography, and measurement of B-type natriuretic peptides and serial serum troponins. Although cardiac contusion can lead to heart failure from valvular injury or cardiac dysfunction, this patient does not have any evidence thus far to support the diagnosis of pulmonary edema.
Patients with pulmonary embolism (Option D) may present with chest pain, dyspnea, and tachypnea. Chest radiograph may demonstrate peripheral, wedge-shaped opacities, but most often the chest radiograph is normal. This patient's clinical course is most consistent with ARDS.
A 40-year-old woman is hospitalized with acute shortness of breath. Medical history is significant for two pregnancy losses in the second trimester. Laboratory testing 3 months ago after her second pregnancy loss was positive for lupus anticoagulant, high titer IgG anticardiolipin antibodies, and high titer IgG anti–β2-glycoprotein antibodies. She is otherwise well and takes no medications. On physical examination, pulse rate is 120/min and respiration rate is 20/min; oxygen saturation is 92% breathing ambient air. Laboratory studies show a positive lupus anticoagulant, high-titer IgG anticardiolipin antibodies, and high-titer IgG anti–β2-glycoprotein antibodies. CT angiography shows a pulmonary embolism in the right main pulmonary artery. Low-molecular-weight heparin is initiated.
Which of the following is the most appropriate long-term treatment?
A. Dabigatran plus aspirin
B. Rivaroxaban
C. Warfarin
D. Warfarin plus inferior vena cava filter
The most appropriate long-term treatment for this patient is anticoagulation with warfarin (Option C).
She meets the criteria for antiphospholipid antibody syndrome (APLAS) based on her vascular thrombosis (acute pulmonary embolism), history of pregnancy morbidity, and laboratory criteria. Pregnancy morbidity that meets the criteria for APLAS includes at least one pregnancy loss after 10 weeks' gestation; at least one premature birth before 34 weeks' gestation because of eclampsia, preeclampsia, or placental insufficiency; or three or more unexplained consecutive spontaneous abortions before the 10th week of gestation. APLAS evaluation includes the anticardiolipin antibodies, anti–β2-glycoprotein antibodies, and lupus anticoagulant; for APLAS diagnosis, laboratory findings must include medium- or high-titer antiphospholipid antibodies on two or more occasions at least 12 weeks apart. This patient has high-risk APLAS based on triple positivity for the lupus anticoagulant, anticardiolipin antibodies, and anti–β2-glycoprotein antibodies on two occasions measured 12 weeks apart.
Emerging data from systematic reviews indicate that the use of direct oral anticoagulants, such as dabigatran, are not as effective as warfarin in the prevention of recurrent thrombotic events in patients with high-risk APLAS. Aspirin is often added to anticoagulation in patients with arterial thrombosis and in patients with a pre-existing indication for aspirin therapy such as cardiovascular or cerebrovascular disease. This patient should be treated with warfarin, not dabigatran, and has no indication for the addition of aspirin therapy (Option A).
Warfarin is the preferred anticoagulant in patients with high-risk APLAS because clinical trials have demonstrated an increased risk of thromboembolic events with rivaroxaban compared with warfarin (Option B).
Guidelines do not recommend dual therapy with an inferior vena cava filter and anticoagulation for patients with deep venous thrombosis (DVT), even for patients with proximal DVT and significant preexisting cardiopulmonary disease, as well as for patients with pulmonary embolism and hemodynamic compromise (Option D).
A 48-year-old man is evaluated for a 25-year history of headaches that have become increasingly frequent. Headaches previously occurred four to five times per month but now occur 16 to 20 times per month and last 12 to 24 hours. The headaches are bilateral, throbbing, moderate in intensity, aggravated by physical activity, and accompanied by photophobia and phonophobia. He has had no other associated symptoms. The patient takes amitriptyline and sumatriptan for the headaches and has been taking sumatriptan four to five times weekly for the past 3 months. Physical examination findings, including vital signs, are all normal. A brain MRI with contrast is normal.
Which of the following is the most appropriate next step in treatment?
A. Begin butalbital
B. Begin verapamil
C. Discontinue amitriptyline
D. Discontinue sumatriptan
The most appropriate next step in management is to discontinue sumatriptan (Option D).
The patient has a history of headaches meeting criteria for episodic migraine and has likely now developed medication overuse headache (MOH). MOH is defined as headache occurring on at least 15 days per month in a patient with a pre-existing headache disorder exposed to regular overuse for more than 3 months of one or more drugs taken for acute and/or symptomatic treatment of headache. Use of triptans, ergot alkaloids, opioids, or combination analgesics for 10 or more days per month or simple analgesics for 15 or more days per month constitutes medication overuse. Affected patients often report daily or near-daily headache that is refractory to numerous treatments. MOH is more common in midlife, in women, and in persons with high baseline headache frequency. The development of MOH often coincides with the transformation of episodic forms of migraine or tension-type headache (occurring <15 days per month) into their chronic subtypes (15 or more days monthly). Treatment of MOH is to wean overused acute medications, initiate migraine preventive medication, and provide new acute medications limited in use to 10 or fewer days per month.
Guidelines suggest butalbital compounds be avoided in patients with primary headache (Option A). In those with migraine, opioids are associated with a 44% increase and butalbital compounds with a 70% increase in the risk of headache progression. Both medications should be avoided in patients with recurrent primary headache disorders, particularly those already diagnosed with MOH.
This patient's headache history is compatible with migraine and subsequent development of MOH. There is no evidence that verapamil is helpful in either headache disorder (Option B).
Amitriptyline is indicated for the preventive treatment of migraine; there is no need to discontinue this medication in the setting of MOH (Option C). Most preventive options for migraine are rendered less effective or ineffective in the presence of MOH. Resolving MOH often restores the therapeutic benefits of pharmacologic prevention of migraine.
In 2025, this historic figure became the first American elected as pope after the death of Pope Francis
Pope Leo XIV
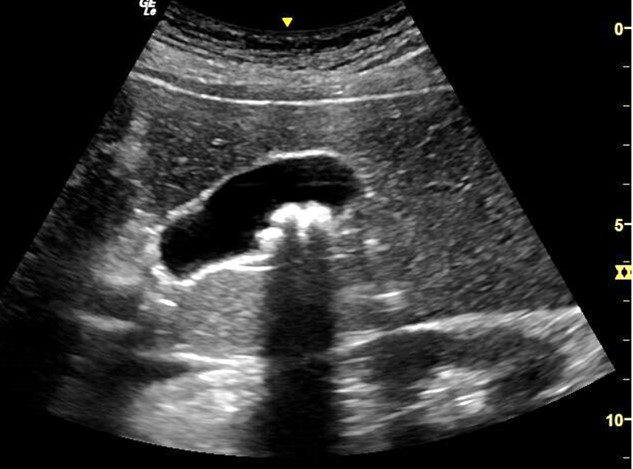
A 55-year-old female presented to the clinic with a chief complaint of right upper quadrant (RUQ) pain for the past 3 months. She also complained that she is not able to tolerate fatty or fried food. There is no history of trauma or surgery. A POCUS right upper quadrant (RUQ) exam was performed. The image above is one of the images obtained during the point-of-care ultrasound exam.
What is the most likely diagnosis?
A. Cholecystitis
B. Cholangitis
C. Cholelithiasis
D. Choledocholithiasis
C. Cholelithiasis
The image shows a gallbladder with multiple hyperechoic stones casting a posterior acoustic shadow in the region of the body of the gallbladder. No stone was seen impacted in the common bile duct. Thus, the possibility of choledocholithiasis is ruled out. The gallbladder wall is not thickened. No pericholecystic fluid seen. Thus, the possibility of cholecystitis or cholangitis is ruled out.
A 24-year-old woman is evaluated for intermittent cough, wheezing, and chest tightness of 1 month's duration. She reports worsening symptoms with exercise and with cat exposure.
On physical examination, vital signs are normal. She has end-expiratory wheezing. Cardiac examination is normal.
On spirometry, FEV1 is 75% of predicted and improves significantly following inhaled albuterol.
Which of the following tests will help predict this patient's responsiveness to inhaled glucocorticoids?
A. Bronchial challenge testing
B. Diffusing capacity for carbon monoxide
C. Fractional exhaled nitric oxide
D. Pulse oximetry
C. Fractional exhaled nitric oxide
Fractional exhaled nitric oxide (FeNO) (Option C) will help predict this patient's responsiveness to inhaled glucocorticoids. Although FeNO should not be used as a diagnostic tool for asthma, it can be used to support this diagnosis in situations in which additional objective evidence is needed. FeNO may be useful if there is uncertainty in choosing, monitoring, or adjusting anti-inflammatory therapies based on history, clinical findings, and spirometry as part of an ongoing asthma monitoring and management strategy. In adults with respiratory symptoms, FeNO levels above 50 ppb correlate with eosinophilic airway inflammation and predict response to inhaled glucocorticoids. FeNO levels below 25 ppb indicate that eosinophilic airway inflammation and glucocorticoid responsiveness are less likely; values between 25 and 50 ppb should be interpreted cautiously. In patients with asthma that is being treated with inhaled glucocorticoids, serial measurement of FeNO may help to monitor patient response to glucocorticoid therapy.
Bronchial challenge testing (Option A) is used to identify bronchial hyperresponsiveness, a diagnostic feature of asthma. This is particularly helpful in patients whose symptoms are suggestive of asthma but for whom other pulmonary function test results are normal. Patients inhale increasing doses of a substance known to induce bronchospasm, such as methacholine or histamine, in a stepwise fashion. This is followed by repeated measurements of FEV1; if FEV1 falls by 20% or more from the baseline value, the test is considered positive.
Diffusing capacity for carbon monoxide (DLCO) measurement (Option B) estimates the amount of gas transfer through the alveolar/capillary unit and is proportional to the surface area of a functional lung. DLCO is measured by inhalation of a gas mixture containing carbon monoxide and helium; the resulting value is corrected for hemoglobin level. DLCO is reduced in conditions in which functioning alveolar capillary units are destroyed, infiltrated, removed, or their function is compromised. Conditions that increase pulmonary capillary blood volume, such as pulmonary alveolar hemorrhage, left-to-right shunt, or asthma, can cause an elevation in DLCO. DLCO measurements do not predict responsiveness to glucocorticoids.
Pulse oximetry (Option D) provides a readily available noninvasive measurement of oxygen-bound hemoglobin in the circulation. A normal hemoglobin saturation measured by pulse oximetry is 95% to 100%, and values below 90% indicate hypoxemia. Pulse oximetry cannot predict responsiveness to glucocorticoids.
A 25-year-old woman is hospitalized for bleeding 1 day following a tonsillectomy. She reports a history of heavy menses since menarche but no spontaneous bruising or muscle or joint hematomas. On physical examination, vital signs and examination findings are normal. Platelet function testing is normal.
Activated partial thromboplastin time (aPTT) 37 s
aPTT with mixing study 26 s
Hematocrit 30%
Platelet count 190,000/μL (190 × 109/L)
Prothrombin time 11 s
von Willebrand factor (vWF) antigen assay 80% (normal)
Ristocetin cofactor activity: vWF antigen >0.7 (normal)
Which of the following is the most appropriate diagnostic test to perform next?
A. Dilute Russell viper venom time
B. Measure factor levels
C. Platelet aggregation study
D. Fibrinogen level
The most appropriate diagnostic test is to measure factor levels (Option B).
The most likely cause of this patient's posttonsillectomy bleeding and history of menorrhagia is factor XI deficiency (hemophilia C), which is rare overall but more common among persons of Ashkenazi Jewish ancestry. Factor XI is part of the intrinsic coagulation pathway, and patients with factor XI deficiency can have isolated activated partial thromboplastin time (aPTT) prolongation. Although patients with severe factor XI deficiency (<20%) are more likely to have a history of bleeding, this can vary and does not tightly correlate with the measured factor XI activity. Patients typically do not experience spontaneous bruising, muscle hematomas, or hemarthroses but tend to have postsurgical bleeding, particularly from sites with high endogenous fibrinolytic activity (dental, nasal, oropharyngeal, genitourinary), or menorrhagia and postpartum hemorrhage. Therefore, even patients with severe factor XI deficiency do not typically require treatment; they may require hemostatic prophylaxis or treatment to control bleeding from surgery. Deficiencies in factors VIII, IX, and XII may also result in isolated aPTT prolongation. However, factor XII deficiency is not associated with a bleeding tendency, and congenital deficiencies in factors VIII and IX are X-linked disorders primarily affecting men, whereas women are asymptomatic carriers. Thus, the most likely diagnosis in this patient is a factor XI deficiency. The mixing study confirms the presence of a factor deficiency by demonstrating complete correction of the aPTT following a one-to-one mix with normal plasma; evaluating factor XI activity would confirm the presence of factor XI deficiency.
The dilute Russell viper venom time is an assay used to confirm the presence of a lupus anticoagulant (LAC), which might be a diagnostic consideration if the prolonged aPTT fails to correct following a one-to-one mixing study (Option A). Most patients with LAC are asymptomatic. Arterial and venous thrombotic events occur in about 30% of patients. Excessive bleeding is not a characteristic of LAC.
Postoperative bleeding may occur in patients with a platelet function disorder; however, platelet dysfunction would be less likely considering the normal platelet count and normal platelet function test results. Additionally, a platelet function disorder would not explain the increased aPTT, so an aggregation study is unhelpful (Option C). Although this patient's history of postsurgical bleeding and menorrhagia is also consistent with von Willebrand disease, the normal von Willebrand factor (vWF) antigen assay and ristocetin cofactor activity:vWF antigen ratio rules out this possibility.
A qualitative defect in fibrinogen (dysfibrinogenemia) or a low fibrinogen level (hypofibrinogenemia) may cause bleeding. Dysfibrinogenemia or hypofibrinogenemia severe enough to cause bleeding, however, would likely result in an increase in the prothrombin time. This patient's prothrombin time is normal, obviating the need to evaluate the fibrinogen level (Option D).
An 18-year-old woman is evaluated for a 5-year history of recurrent headaches. Headaches occur approximately 12 days per month; three of these episodes are severe, occurring without a trigger or premonitory symptoms, and last for a full day, followed by profound fatigue the next day. She describes the pain as bilateral, frontotemporal, steady and throbbing, associated with sensitivities to light and noise, worsening with movement, and causing her to miss 2 or 3 days of school per month. The severe headaches have not responded to ibuprofen or naproxen. The patient also has seasonal allergies. Other medications are fexofenadine and a combination estrogen-progestin oral contraceptive.
All physical examination findings, including vital signs, are normal.
Which of the following is the most appropriate next step in management?
A. Begin oral zolmitriptan
B. Discontinue the oral contraceptive
C. Obtain a brain MRI
D. Substitute loratadine for fexofenadine
This patient has episodic migraine without aura and should be treated with an oral triptan such as zolmitriptan (Option A).
Migraine is thought to be a manifestation of a central nervous system that is biologically hypersensitive and prone to episodes of disabling headache. Once initiated, the migraine attack may possess not only the phases of aura and headache, but also of prodrome and postdrome. Prodromal or premonitory symptoms may precede these other phases by hours or sometimes days and, given that timing, they occasionally may be confused for a triggering influence. After severe attacks, postdrome is quite common and often described as a “headache hangover.” Evidence-based guidelines recommend several simple and combination analgesic agents as first-line therapies for acute migraine. Aspirin administered alone or in combination with acetaminophen and caffeine, ibuprofen, naproxen sodium, and dissolvable diclofenac potassium are all supported by strong evidence. Triptans are migraine-specific selective agonists at 5-hydroxytryptamine 1B and 1D receptors with direct impact on trigeminovascular activation associated with migraine attacks. Guidelines recommend the use of triptans (such as zolmitriptan) in patients with moderate to severe migraine who have not responded to NSAID therapy over a series of at least three migraine attacks. Current evidence suggests that all oral triptans possess nearly similar clinical efficacy.
In the absence of aura, there is no indication to discontinue estrogen-containing oral contraceptives (OCPs) (Option B). There appears to be no significant increase in stroke risk from OCPs in patients with migraine without aura.
In the presence of a stable clinical pattern of migraine and a normal neurologic examination, guidelines recommend against brain MRI (Option C).
Neither fexofenadine nor loratadine have any demonstrated benefit in migraine management, so there would be no indication for switching agents (Option D).
In October 2025, thieves disguised as construction workers used a truck‑mounted lift to break into which national museum and art gallery in France?
Louvre
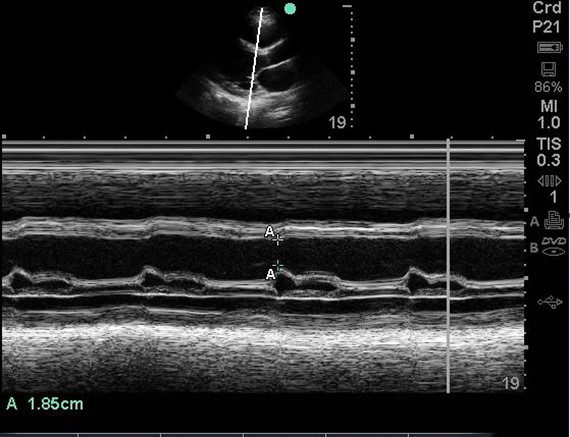
A 75-year-old male with previous medical history of hypertension (HTN), diabetes mellitus (DM) type II and ongoing tobacco use presented with complaint of worsening dyspnea, orthopnea, weight gain and lower extremity edema. The patient also has a family history of ischemic heart disease, cardiovascular accident (CVA).
Temperature - 98.4 degrees Fahrenheit/36.9 C, HR - 68 BPM, BP – 137/84 mm Hg, RR – 20 per minute. EKG shows – Q waves in inferior and lateral leads. No ST-T changes.
Physical exam is remarkable for jugular vein distention (JVD), rales / crackles in lung bases, 1+ pitting edema up to the lower leg (bilateral). A POCUS cardiac exam was performed. An E-Point Septal Separation (EPSS) measurement of 1.85cm was obtained.
What is the approximate ejection fraction (EF) based upon the EPSS measurement?
A. The EF is approximately 50%
B. The EF is approximately 30%
C. The EF is approximately 70%
D. The EF is approximately 10%
B. The EF is approximately 30%
A study using MRI data by Jay R Silverstein et al proposed that the mitral valve E point septal separation (EPSS) can be used to quantify LV ejection fraction on a continuous scale. They proposed the following formula to estimate EF on a continuous scale rather than just saying normal or reduced EF.
LVEF = 75.5 – (2.5 x EPSS [millimeters])
Based upon the above equation, the EPSS is 29.25%. It agrees with the chart above as well. Keep in mind that the formula is for calculations using MRI data. But we can get an approximate idea. Note that this is still an approximate estimation of the EF. True EF can only be extrapolated from volumetric data.