What is an alloy?
A solution of metals.
The ____________ is dissolved within the ___________ to make a Solution.
solute, solvent
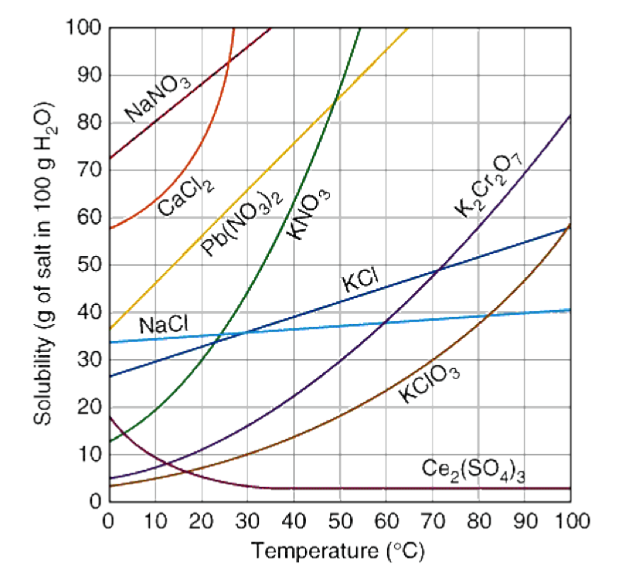
Which solute is least soluble at 10 ⁰C
KClO3
This is the universal solvent.
What is water?
TRASHKETBALL SHOT!!!
x1 x2 x3
done
This vocab word refers to the amount of solute that can be dissolved in a volume of solvent to make it saturated.
Solubility

Which solute is the most soluble at 10 ⁰C?
NaNO3
TRASHKETBALL SHOT!!!
x1 x2 x3
done
If you were making Lemonade, what would be the solvent?
Define the word insoluble.
A substance that cannot dissolve in a solvent

How many grams of K2Cr2O7, are soluble in 100 g of water at 90 ºC?
70 g
Air
Wood
Salt
PURE water
Air
Name three things that will increase the rate at which a solute dissolves in a solution.
Heat it
Crush it - smaller particles (more surface area)
Stir it
If a solution contains as much solute as a solvent can dissolve it is this.
Saturated

When 30 grams of KClO3 is dissolved in 100 grams of water at 60 ºC, the solution can be correctly described as what?
Unsaturated, Saturated, Supersaturated
Supersaturated
What is the predominant relationship between temperature and solubility?
Positive, direct correlation
You are given a small beaker of solution at room temp. You add a bit of solute to the solution and it dissolves. Was the solution unsaturated, saturated, or supersaturates?
Unsaturated
What is the term that describes a solution that has more solute dissolved than it should be able to at that temperature?
Supersaturated

You have a solution of K2Cr2O7 containing 10 grams of solute at 50⁰C. How many additional grams of solute must be added to the solution to make the solution saturated?
20 g
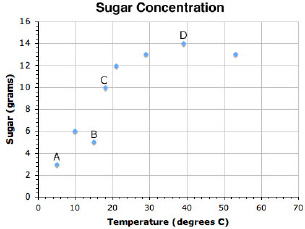
While making lemonade. You reference this solubility graph. What temperature would be the BEST to heat the tea when adding sugar, if you prefer your tea as sweet as possible?
D; 39 ºC