Balanced equation / mole ratio
How can I go from grams of a substance to moles of a substance?
Divide moles by the molar mass
What's the first step from converting moles of a substance to mass of another?
A reactant that does not get used up in the reaction
Excess reactant
How many moles are in 23 grams of H2O?
1.28 moles
Consider this equation:
4 Ti + 3 O2 → 2 Ti2O3
What's the mole ratio of Ti and O2 in the balanced equation.
4:3
A reactant that DOES get used up in the reaction
Limiting reactant
Consider the following equation:
CH4 + 2 O2 → CO2 + 2 H2O
160 grams of CH4 can form how many moles of CO2?
10 moles CO2
How many grams are in 18 moles of CO2?
792 grams
Recipe: 2 slices of bread + 3 slices of meat + 1 slice of cheese --> 1 Sandwich
Inventory: 8 slices of bread, 9 slices of meat, 5 slices of cheese
What's your limiting reactant?
Meat
Consider the following equation:
CH4 + 2 O2 → CO2 + 2 H2O
160 grams of CH4 can form how many moles of H2O?
20 moles of H2O
Consider this reaction: 4 Ti + 3 O2 → 2 Ti2O3
How many grams of Ti2O3 are formed when 24 moles of Ti react?
1,728 grams
Recipe: 2 slices of bread + 3 slices of meat + 1 slice of cheese --> 1 Sandwich
Inventory: 8 slices of bread, 9 slices of meat, 5 slices of cheese
How many sandwiches could you make?
3 Sandwiches
What type of reaction is
CH4 + 2 O2 → CO2 + 2 H2O
Combustion
Consider this reaction: 4 Ti + 3 O2 → 2 Ti2O3
What mass of Ti2O3 will be produced by the reaction of 24.0 moles of O2?
2304 grams
Consider
H2 + Cl2 --> 2 HCl
If you have 6 moles of H2 and 4 moles of Cl2, how many moles of HCl can be formed?
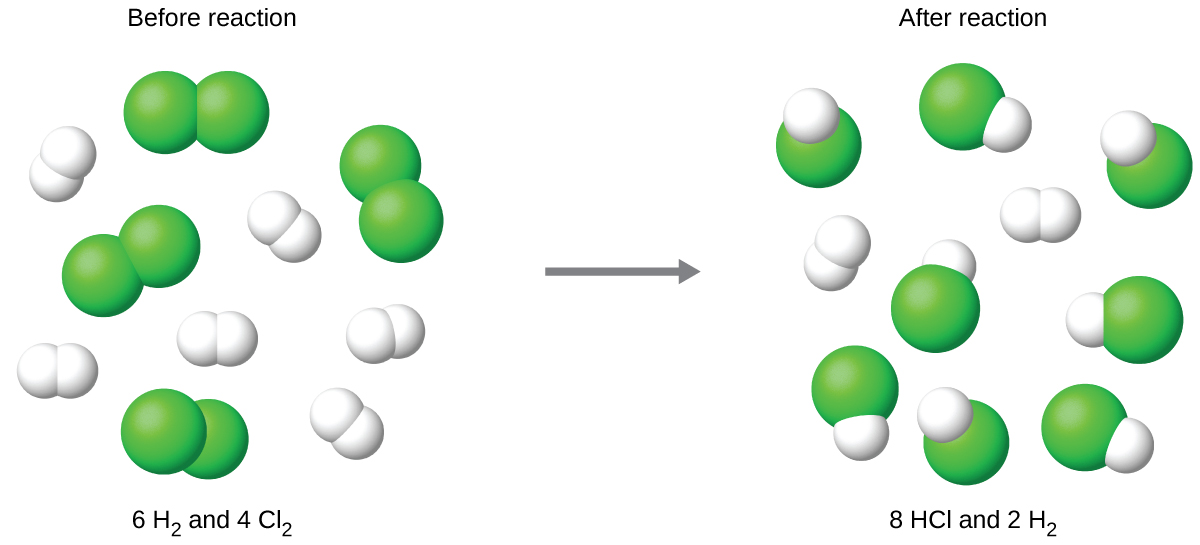
8 mol of HCl