Increases across a period
Atomic number/ Nuclear charge (number of protons)
What is oxidation and reduction?
Oxidation is loss of electrons
Reduction is gain of electrons
Electrons in the outer most energy level.
Valence electrons
What is the difference between empirical and molecular formulas?
Empirical is the simplest whole number ratio while molecular is not
Draw out the skeletal formula for 2-methylbutane.

Increases down a group
Energy levels
What increases during oxidation and decreases during reduction?
Oxidation state
A material whose physical properties resemble metals and chemical properties resemble nonmetals.
Metalloids
What part of the skeletal formula would represent a carbon?
The bend and/or end
Why dont noble gases generally have electronegativity?
They are stable and dont need to attract electrons
Increases across a period and decreases down a group (List all)
Electron affinity, ionization energy, electronegativity
What is oxygens oxidation state?
-2
A more reactive element replaces a less reactive one.
Displacement reaction
What is the iupac suffix for an ester?
Anoate
Name:
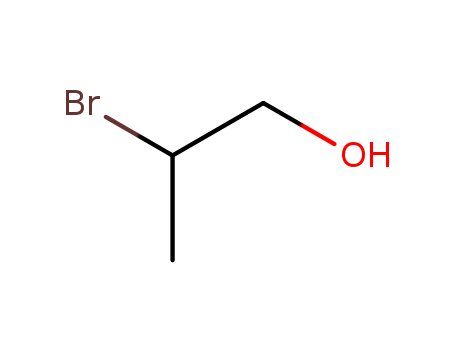
2-bromopropan-1-ol
Decreases across a period and increases down a group
Atomic radius and ionic radius
This type of agent donates electrons and is oxidized
Reducing agent
Measure of attraction between incoming electron and the nucleus.
Electron Affinity
What is the definition of a homologous series?
A family of compounds which members differ by a common structural unit (ex. CH2)
Which element has the lowest electronegativity value?
Potassium Iodine Flourine Hydrogen
Potassium
Why does halogen reactivity decrease down the group?
Larger atoms gain electrons less easily
Oxidation and reduction occur in what type of reaction?
Redox reactions
Can act as both an acid and a base.
Amphoteric oxide
Define an optical isomer.
Steroisomers that are mirror images of one another
A student places sodium and potassium in water. Explain which reacts more vigorously and why.
Potassium reacts more vigorously because it is lower in Group 1, has lower ionization energy, and loses its valence electrons more easily.