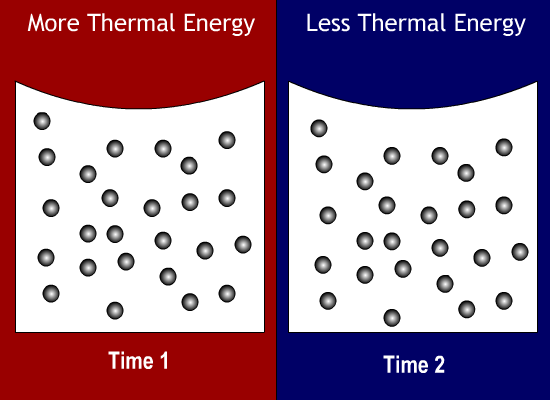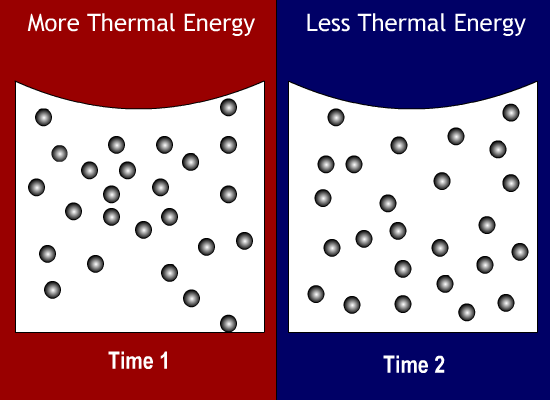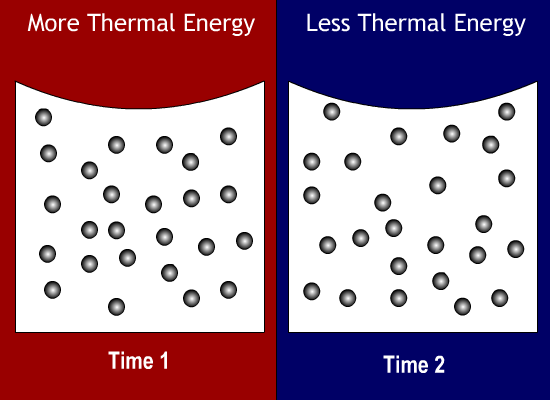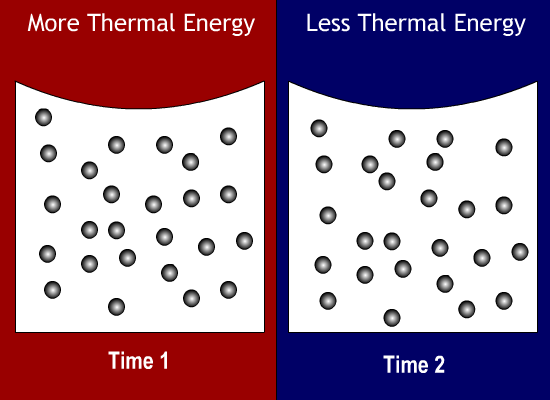
What state of matter has a definite shape and a definite volume?
What is Solid?
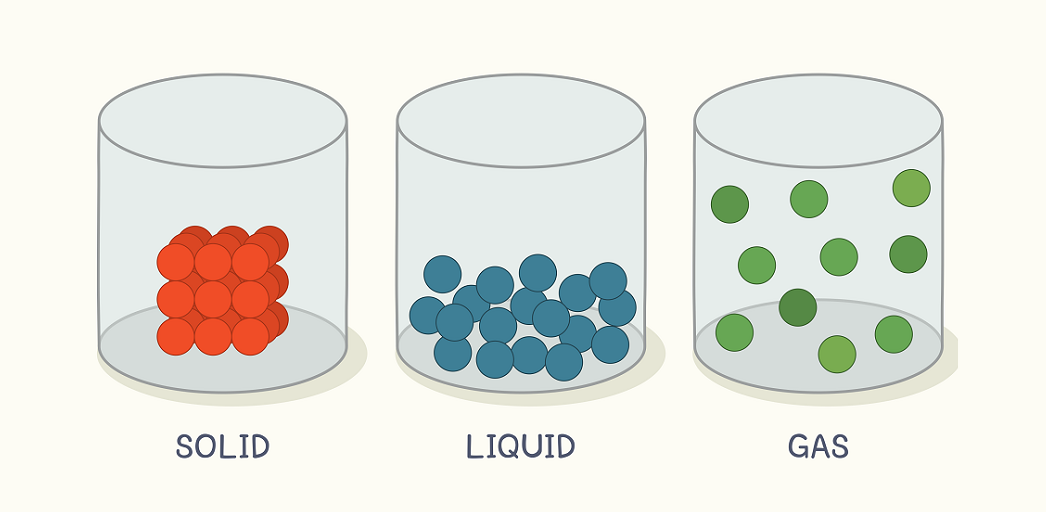
What are the three states of matter?
Solids, liquids, and gases
What is it called when a solid changes to a liquid?
melting

What is it called when a liquid changes to a solid?
Freezing

When energy is added to a substance, the particles will move (faster/ slower)?
faster
A substance that has neither a definite shape nor a definite volume.
Gas
In a solid, particles do not move around each other, but they do
Vibrate in place

The change in state from a gas to a liquid.

Condensation

What is called when a liquid changes to a gas?
Vaporization

When a liquid is freezing into a solid, thermal energy is...(added/ taken away)
taken away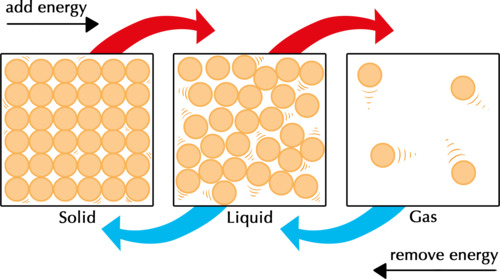
This state of matter has a definite volume but no definite shape
Liquid
What type of solid does not have particles arranged in a regular pattern and does not melt at a specific temperature? Example: butter
:max_bytes(150000):strip_icc()/sticks-of-butter-photo-by-twoellis-GettyImages-149134517-resized-3911123142a141eca2340a4bb63e0869.jpg)
Amorphous solid

What is vaporization that only takes place on the surface of the liquid?
evaporation

When a substance is cooled, its thermal energy will do what?
Decrease
During sublimation (the change from solid to gas) thermal energy is (added/ taken away)
added
What state of matter is milk?

Liquid
Helium is an example of which state of matter?
Gas
What is the change in state when a solid changes directly to a gas?
sublimation

What is the temperature at which a substance changes from a liquid to a solid?
freezing point
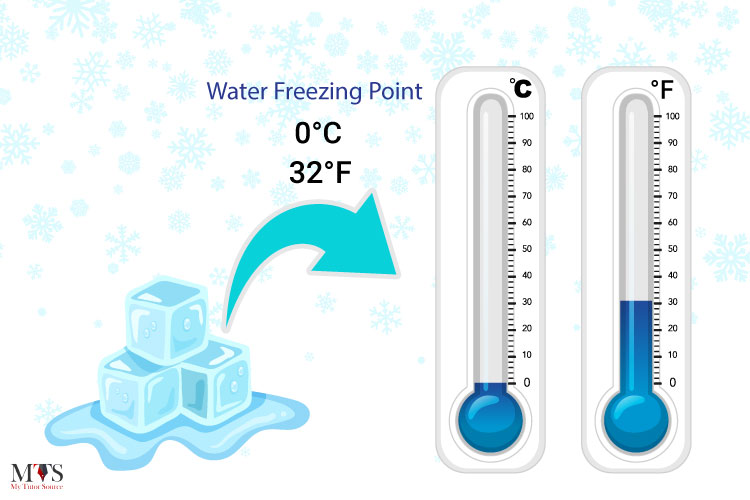
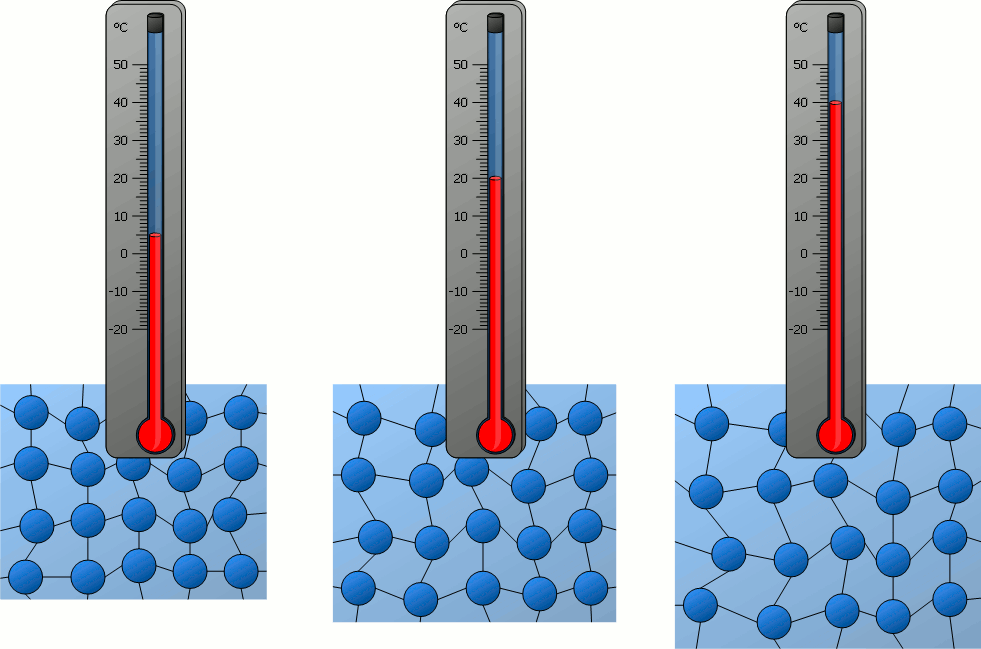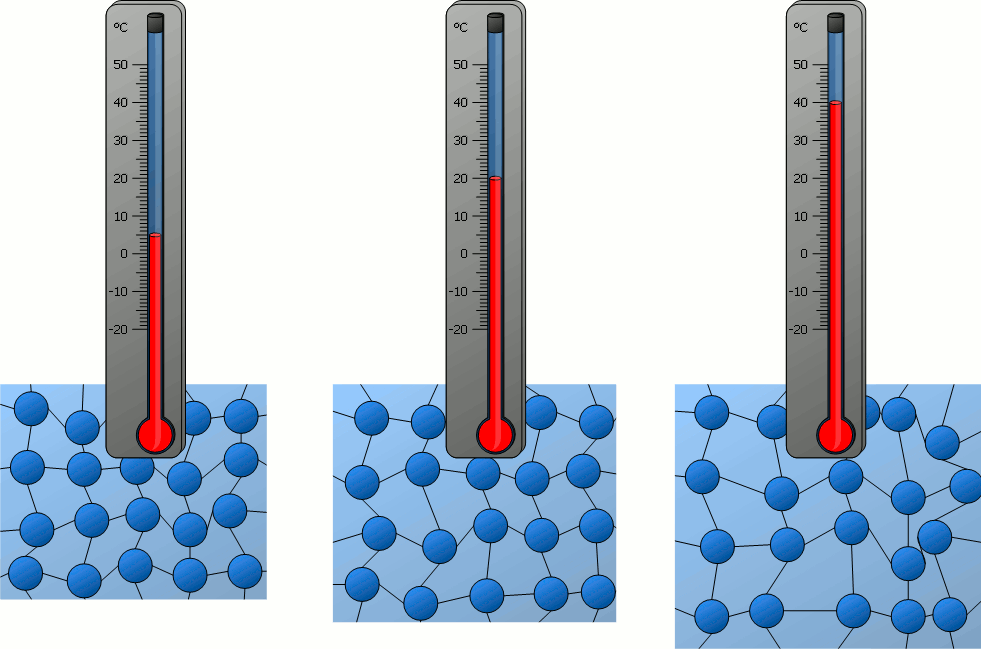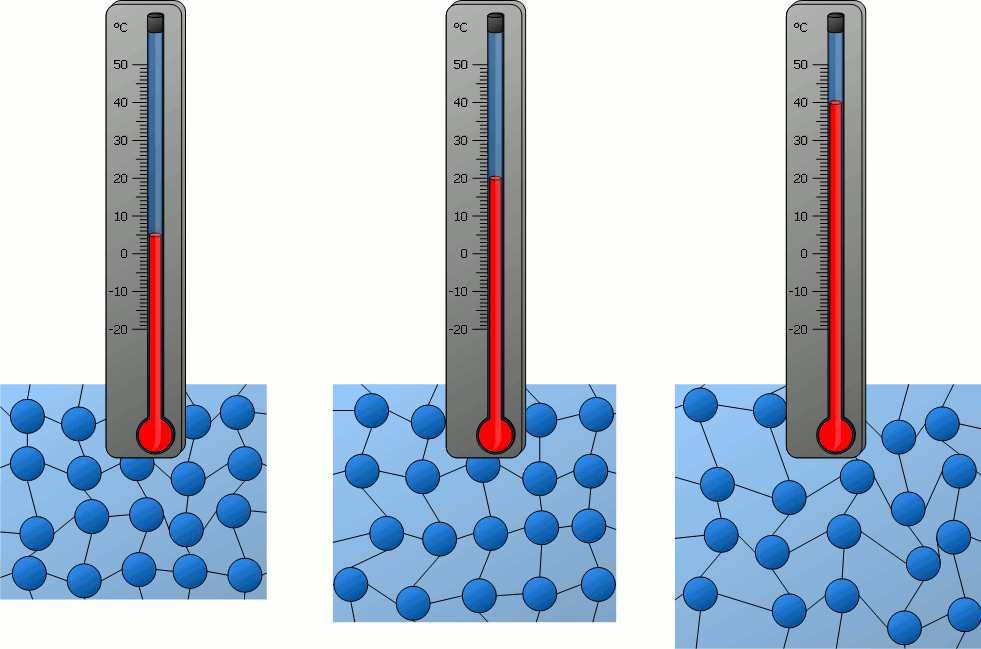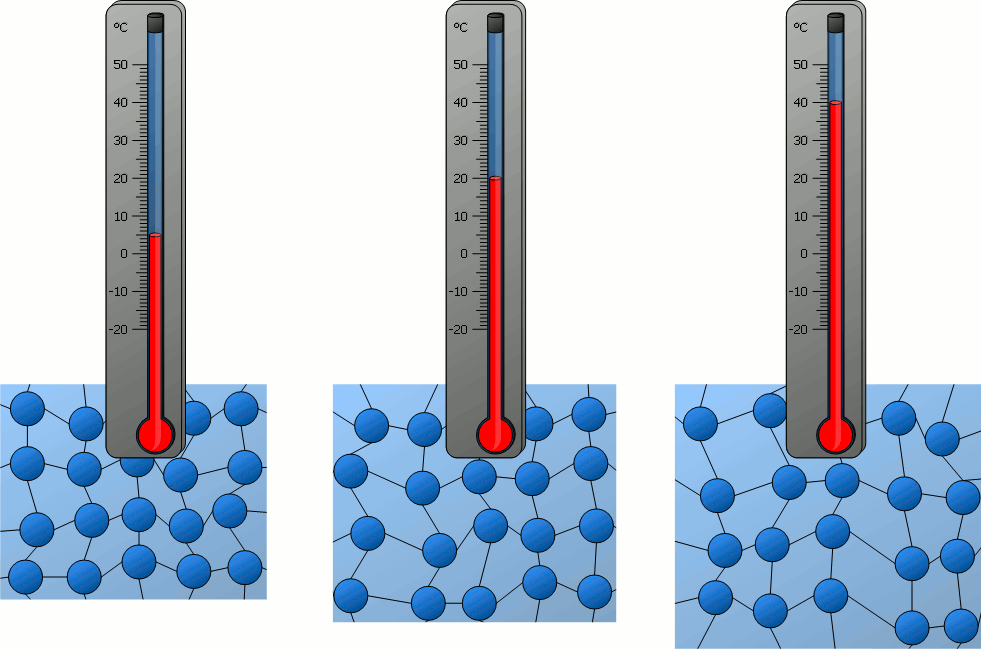
What is temperature? What does it measure?
Temperature is a measure of the average kinetic energy of the particles in an object or substance. It measures how quickly the particles are moving.

Sugar is considered a crystalline solid because its particles are
Arranged in a regular pattern
What is the property of liquids that causes their surface to behave like a thin, elastic film?
:max_bytes(150000):strip_icc()/Surface-Tension-58c6c2365f9b58af5c534f71.jpg)
 Surface tension
Surface tension
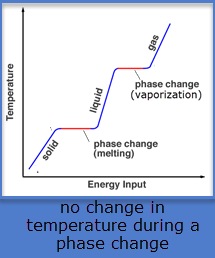
What happens to the temperature of a substance during the phase change (change of state)? (melting, freezing, vaporizing, condensing)
Does it increase? Decrease? Stay the same?

Stays the same
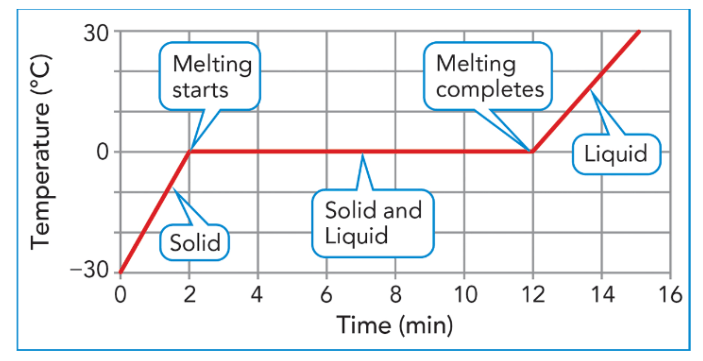
What do you notice about the vaporizing point and condensing point on this graph?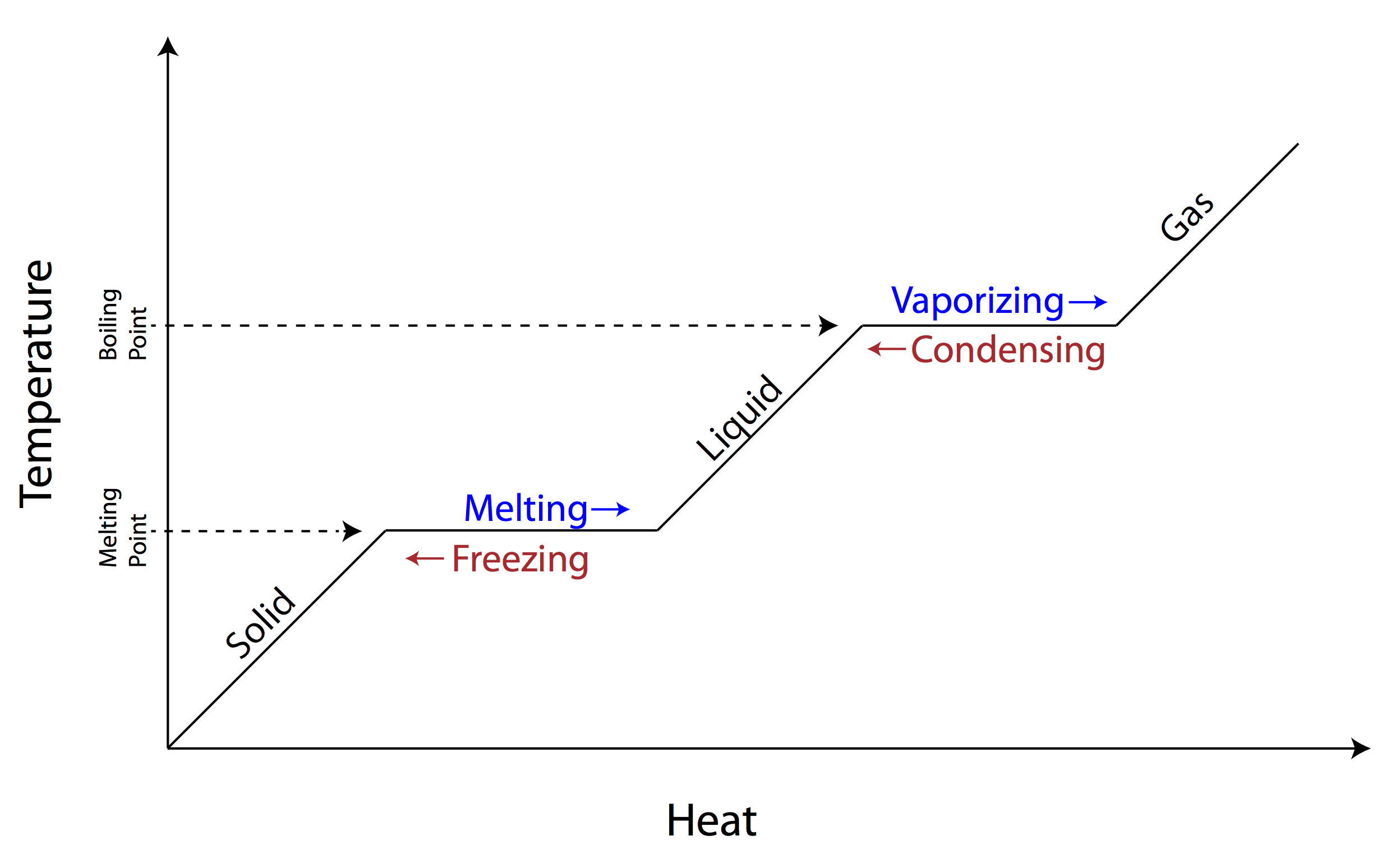
Answers may vary:
*Vaporizing (boiling) and condensing points are the same, because they are the temperatures at which change occurs.
*Temperatures remain the same as substances change state.
What is the total kinetic and potential energy of all the particles in an object or substance?

thermal energy