The change of state from a solid to a liquid
Melting
The correct label for A, C, and E
A - Solid
C - Liquid
E - Gas
What is the point at 5.2 atm and 216.6 K called?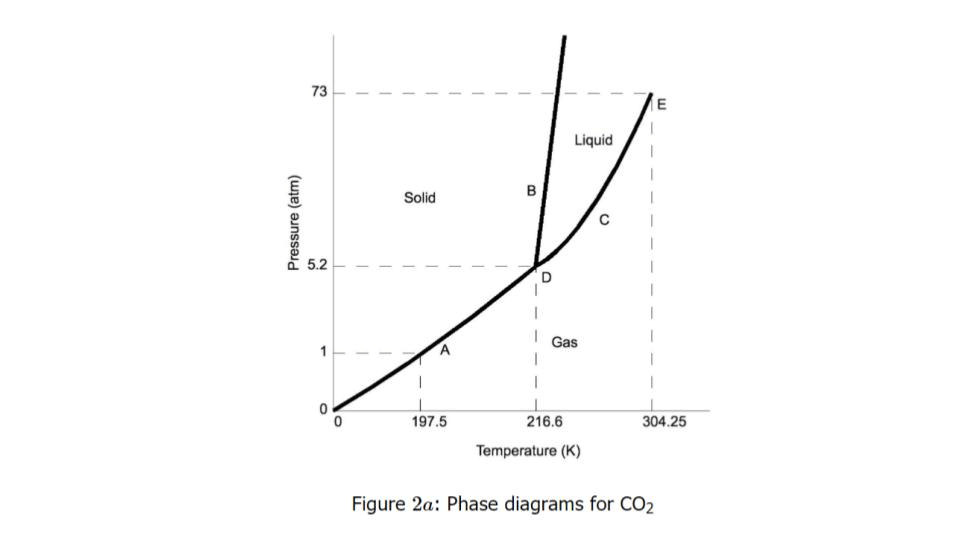
Triple point
What is specific heat?
Quantity of heat needed to raise the temperature of a substance
Give the difference between endothermic and exothermic
Endothermic - cold, absorbs energy
Exothermic - hot, releases energy
Transfer of heat through the sun or fire
Radiation
The Boiling point for the Heating & Cooling Curve 
1465
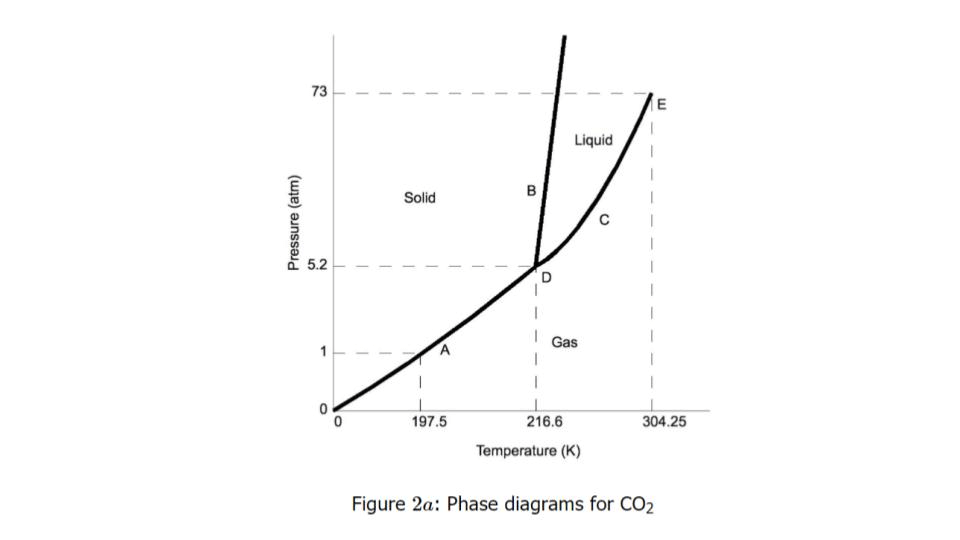 At 5.2 atm and 216.6 K, what states of matter are present for CO₂?
At 5.2 atm and 216.6 K, what states of matter are present for CO₂?
Solid, liquid, and gas
Look at the table provided above. Which substance would be the hardest to heat up, and why?
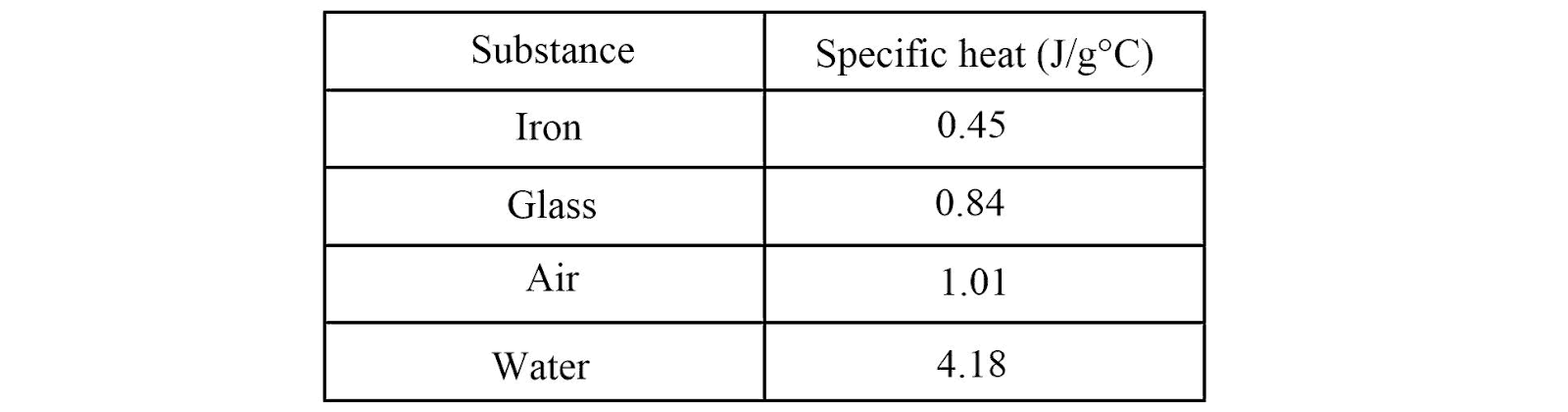
Water, because it has the highest specific heat
What would likely happen if you were to touch the flask in which an endothermic reaction was occurring?
It would feel cold
Device used to measure the heat absorbed or released during a chemical or physical process
Calorimeter
The approximate melting point for the Heating & Cooling Curve below
801
At a constant temperature of ~300 K, if pressure decreases from 70 atm to 1 atm, what phase change will CO₂ undergo?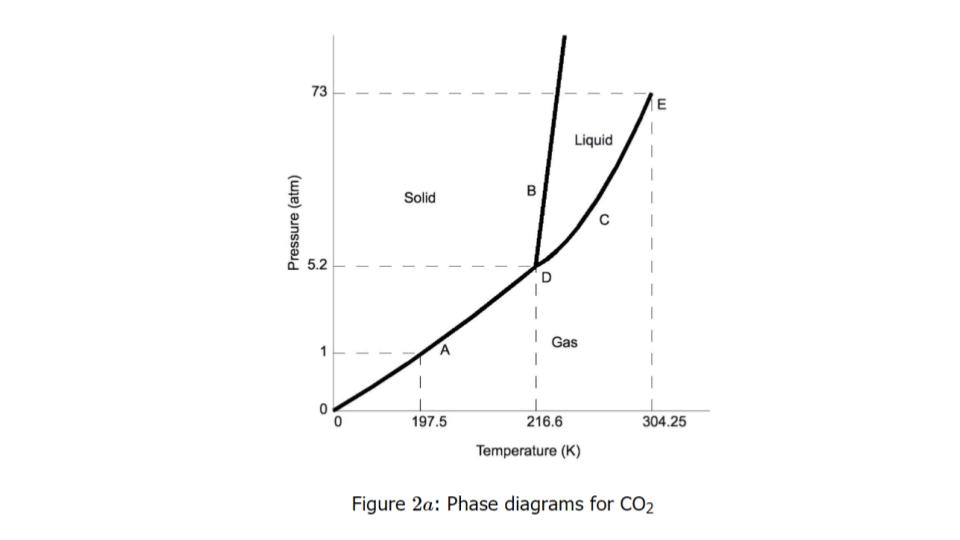
Liquid → Gas
If you wanted to slowly heat a cup of liquid without it changing temperature quickly, which substance from the table would you choose, and why?
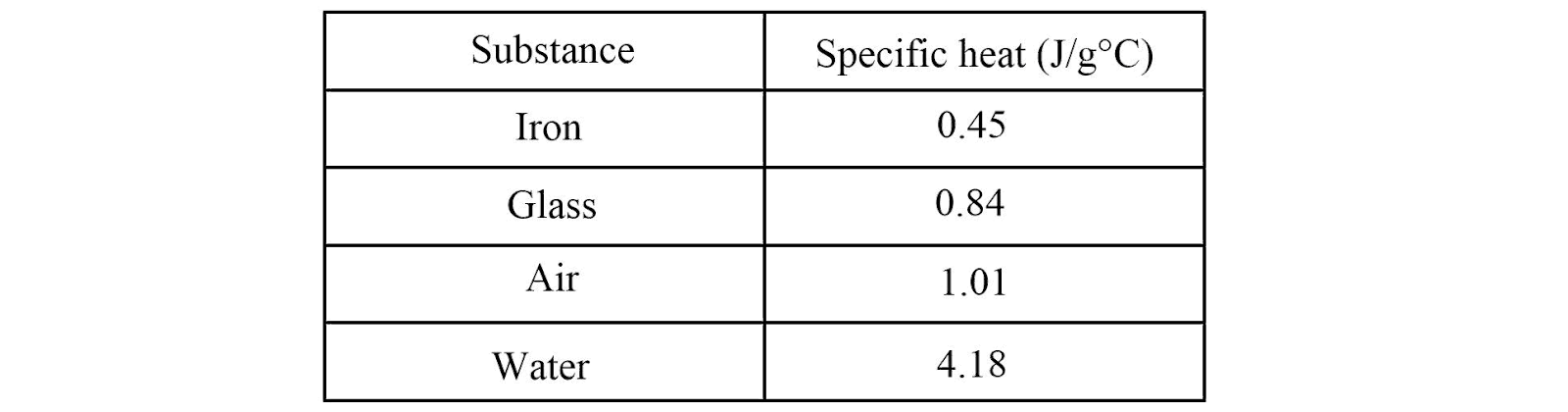
Water, because it has a high specific heat and resists temperature change
A piece of metal is heated, then submerged in cool water. Which statement below describes what happens?
a. The temperature of the metal will increase.
b. The temperature of the water will increase.
c. The temperature of the water will decrease.
d. The temperature of the water will increase and the temperature of the metal will decrease.
D.
Give the difference between conduction and convection
Conduction - Transfer of heat through direct contact
Convection - Transfer of heat through the movement of heated particles (no touching)
The correct label for B and D for the Heating & Cooling Curve below
B - Melting
D - Boiling/Evaporation
 As the pressure increases from 1 atm to 70 atm at a constant temperature of 197.5 K, what happens to the particles of CO₂?
As the pressure increases from 1 atm to 70 atm at a constant temperature of 197.5 K, what happens to the particles of CO₂?
Particles slow down and get closer together
How much heat is required to raise the temperature of 10 g of aluminum by 10℃? (Specific heat of aluminum = 0.21 J/g·℃)
21 J
During a phase change, the temperature of a substance ____.
a. Increases
b. remains constant
c. Decreases
d. may increase or decrease
B.
Give the definitions of Heat and Temperature
Heat - Amount of thermal energy possessed by a substance
Temperature - Measure of average kinetic energy of the particles in a sample
Label H, J, K, and I
J & K = Exothermic
If you want CO₂ to sublimate rather than melt, which conditions must you use?
Pressure below 5.2 atm and temperature below 216.6 K
Look at the table provided above. If you apply the same amount of heat to 100 g of iron and 100 g of water, which one will experience a larger temperature change?

Iron, because it has a lower specific heat
Boiling is a ________________ process since energy must be _______________