Temperature
Rank the following IMFs from strongest to weakest force: Dipole-Dipole, LDFs, H-Bonds
H-Bonds > Dipole-Dipole > LDF
Define specific heat capacity.
The amount of energy it takes to change the temperature of a substance by a degree celsius.
The term (type of rxn) used to describe energy being released from the systems to the surroundings.
Exothermic Reaction!
When a gas turns into a liquid we call that process...
Condensation!

When two objects that are different temperatures reach the same temperature over time, we call that final state...
Thermal Equilibrium
What intermolecular force(s) will SO2 experience?
LDFs and Dipole-Dipole

Why does water have a higher specific heat capacity than most other substances? Explain.
Water has a higher specific heat capacity compared to most other substances due to hydrogen bonding properties. Water molecules can form extensive hydrogen bonds with neighboring molecules, leading to a higher energy requirement to break these bonds and increase the temperature of water. This means that water can absorb or release a larger amount of heat energy for a given change in temperature compared to other substances with weaker intermolecular forces.
This property of water makes it an excellent regulator of temperature in various environments, such as providing a stabilizing effect on climate and maintaining favorable conditions for life in aquatic ecosystems.
Are the following reactions exothermic or endothermic?
1. Water solidifies into ice in a freezer
2. Water evaporates from a glass left on a table
3. Dew condenses on grass overnight

1. Exothermic (freezing)
2. Endothermic (evaporation)
3. Exothermic (condensation)
Label ALL points on the following phase diagram.
A = Solid
B = Liquid
C = Gas
D = Critical Point
E = Triple Point
The law that states that energy cannot be created or destroyed
Law of Conservation of Energy
Which substance will have a larger vapor pressure? Why?
Br2 or NH3?
Br2 will have a larger vapor pressure because of its weaker IMFs (dispersion forces). The strength of the IMFs are inversely proportional to vapor pressure, meaning as the strength of the IMF increases, the vapor pressure decreases.
This is because vapor pressure only applies to substances in their gaseous state. Substances with weak IMFs require less heat (energy) to turn into a gas (change phases).
After a cold ice cube is placed on a 25.0 g aluminum block, the temperature of the block decreases by 8.00oC. How much heat does the aluminum block lose in Joules?
The specific heat capacity of aluminum = 0.897 J/goC
q = -179 J
q = (25.0 g) x (0.897 J/goC) x (-8.00 oC) = -179.4 J
Draw the reaction coordinate diagram for an endothermic reaction.
Label the reactants, products, and change in energy (H)

Which letter represents a substance that is a solid, liquid, and gas at the same time? What is that point called?
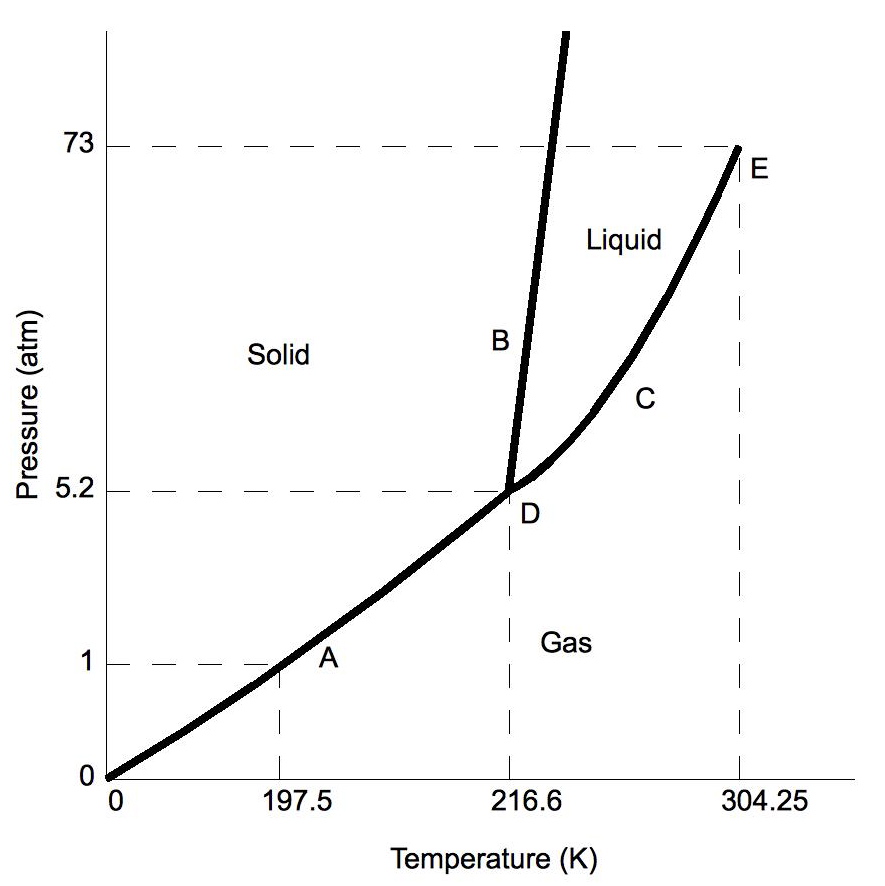
D, the Triple Point!
The pressure at which the rate of evaporation equals the rate of condensation in a sealed container.
Vapor Pressure
Which substance would experience a larger IMF? Why?
H2O or HF?
H2O would because it has more electrons which means more H-bonds and thus an overall larger net force.
(larger molecular weight)
DAILY DOUBLE!
Find the specific heat capacity of iron when 150.0 g of the iron was heated to 125.0oC and placed in a calorimeter, which contained 75.00 g of water. The water temperature rose from 21.7oC to 40.0oC.
ciron = 0.450 J/goC
2 NO (g) + O2 (g) --> 2 NO2 (g)
1. What is the overall change in enthalpy of this reaction?
2. Endothermic or Exothermic?
ΔHof of NO = 90.3 kJ/mol
ΔHof of O2 = 0 kJ/mol
ΔHof of NO2 = 33.2 kJ/mol
(2 x -33.2 kJ/mol) - [(2 x 90.3 kJ/mol) + (0 kJ/mol)]
ΔH = -114.2 kJ
Which phase transition is represented if a substance's pressure decreased from 5.2 atm to 0.5 atm at 200 K?

Sublimation
The scientific method of measuring the change in energy of a system. Such as measuring the amount of heat released or absorbed during a chemical reaction.
Calorimetry
Rank the following compounds from highest to lowest boiling point:
CO2, CH4, KOH, HBr
KOH > HBr > CO2 > CH4
1. Rank by IMF strength
2. Rank by molecular weight
A 55.1 g piece of metal is heated to 45.1oC and placed into a cup containing an unknown amount of water at 20.0oC. The final temperature of the water and metal is 22.3oC.
How much water in grams was the metal placed in?
*The specific heat capacity of the metal is = 2.75 J/goC
mwater = 359 g
CH4 (g) + 2 O2 (g) --> CO2 (g) + 2 H2O (l)
1. What is the overall change in enthalpy of this reaction?
2. Endothermic or Exothermic?
ΔHof of CH4 = -74.8 kJ/mol
ΔHof of O2 = 0 kJ/mol
ΔHof of CO2 = -393.5 kJ/mol
ΔHof of H2O = -285.8 kJ/mol
[(-393.5 kJ/mol) + (2 x -285.8 kJ/mol)] - (-74.8 kJ/mol)
ΔH = -890.3 kJ
Which phase transition is represented if a substance's temperature increased from 230 K to 350 K at 65 atm?

Vaporization!