What elements are found in carbohydrates?
Carbon, Hydrogen, and Oxygen
What elements are found in lipids?
Carbon, hydrogen, and oxygen
What are the monomers of proteins?
Amino acids
Enzymes are biological ________.
Catalysts
What is the charge of an electron?
Negative
What are two properties of water?
Density, polarity, high specific heat capacity, high latent heat of vaporization, cohesion, and adhesion
1. Draw and label a phospholipid with the following terms:
Head, Tail, hydrophilic, hydrophobic
2. What do phospholipids make-up in our cells?
What is the function of carbohydrates?
Short term energy storage
Which of the following is not a type of lipid?
a. Fatty Acid
b. monosaccharide
c. wax
d. triglyceride
b. monosaccharide
What are two functions of proteins?
Signaling and transporting molecules, defense (antibodies), structure, contractile motions, digestive enzymes, and storage
Competitive inhibitors bind to what part of the enzyme?
Active site
What is the charge of a proton?
Positive
Cohesion is:
same molecules are attracted to one another
What are polysaccharides?
Long chains of monosaccharides
What is the function of lipids?
Long term energy storage
Which of the following is not a type of secondary structure in proteins?
Alpha helix
Beta pleated sheet
Subunit
3. Subunit
Enzymes are a type of what macromolecule?
Proteins
What is the charge of a neutron
Neutral (0)
Why does ice float on top of liquid water?
Ice is more dense than liquid water so it floats to the top
The water molecules in ice are more closely packed together than they are in liquid water
The bonds between water molecules are weaker in ice than they are in liquid water
Ice has a crystalline structure with more space between the water molecules than in liquid water
4. Ice has a crystalline structure with more space between the water molecules than in liquid water
Which type of reaction is used to form disaccharides from monosaccharides?
Dehydration Synthesis
What is the name of this lipid?
Triglyceride
What is the name of this protein molecule?
Polypeptide chain
Enzymes increase the rate of reactions by lowering the __________ __________.
Activation energy
Covalent bonds are between what two molecules?
Nonmetal and Nonmetal
What type of bond is shown in the water molecule?
Hydrogen bond
Which type of reaction is used to break down polysaccharides and disaccharides into monosaccharides (water is added to)?
Hydrolysis
What is the primary function of lipase enzymes?
A. breaking down fats
B. breaking down proteins
C. breaking down carbohydrates
D. breaking down starches
A. breaking down fats
A tertiary structure of proteins gets folded into a _____ shape.
3D shape
The graph below shows the effect of pH on enzyme activity.
At which pH value(s) are the enzymes catalyzing the most substrate?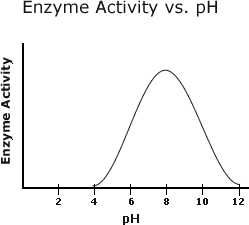
pH of 8
How many valence electrons are in this atom?
7 valence electrons
The properties of water make it essential for supporting life on earth. One of its properties helps it act as a buffer for temperature changes, which stabilizes the environment for creatures that live in water. Which property is this?
A. High specific heat capacity
B. Density
C. Polarity
D. High latent heat of vaporization
A. high specific heat capacity