
What kind of change is this?
Physical
10 cm3 is equal to how many mL?
1 cm3 = 1mL
10 mL
What is the independant variable?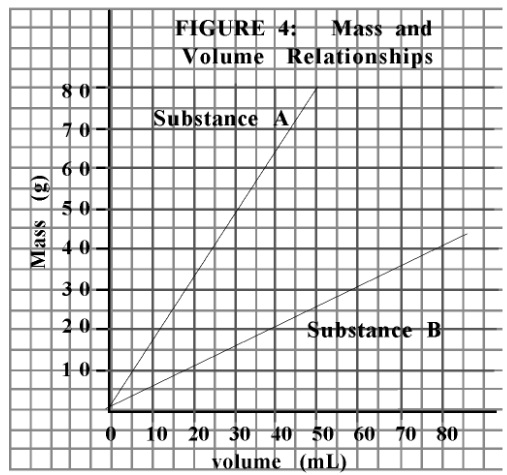
Volume (mL)
list the prefixes from largest to smallest. Hint: There are 6
Kilo
Hecto
Deka
deci
centi
milli
What is the formula for Density?
D=M/V
What kind of change is this?

Chemical
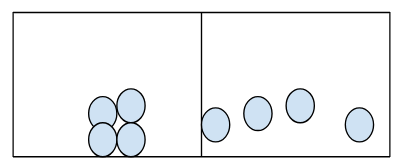
Draw a particle picture for ice melting
What are the units for slope?
g/mL
:max_bytes(150000):strip_icc()/meniscus01-58b5b2c03df78cdcd8ab8299.png)
Give the correct measurement for this instrument.
24.0 -24.3 mL
If you blow up a balloon and then let out a quarter of the air from the balloon, what happens to the density of the balloon from when it was the original size to when the size was reduced?
It stays the same

What kind of change is occuring
Chemical
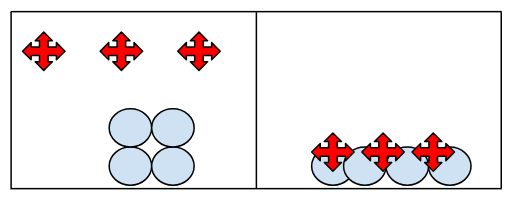
Steel Wool is heated in a flame and reacts with oxygen in the air. The steel wool gains mass. Draw a particle picture.
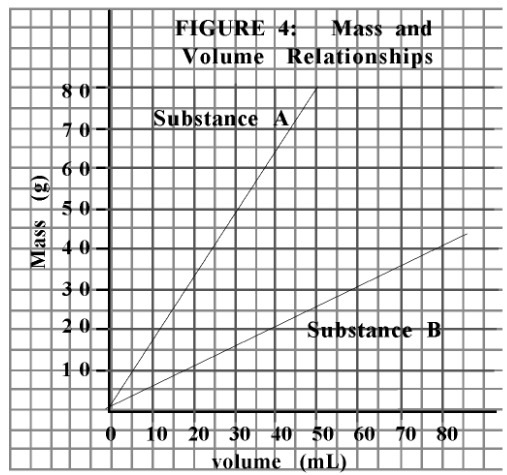
What is the slope of substance A?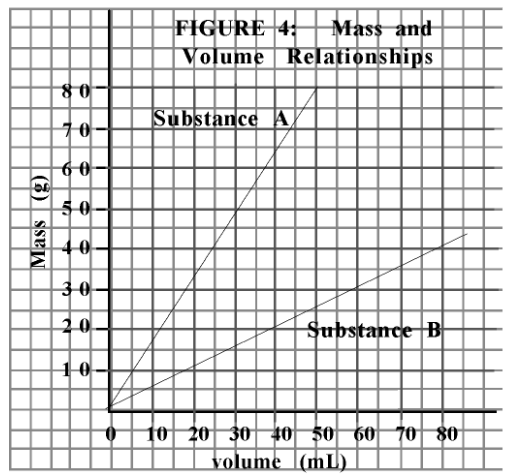
1.6 g/mL

Find the length of the screw.
5.11 cm
What is the density of a piece of wood that has a mass of 25 grams and a volume of 294 cm3? Would the piece of wood sink or float on water?
0.85 g/cm3 , yes it will float on water

What kind of change is this?
Physical
You are given a block of silver that has a density of 10.50 g/cm3. You measure the dimensions of the block to be 2 cm x 4 cm x 3 cm. How many grams does the block weigh?
252 g
Which substance would float on water?
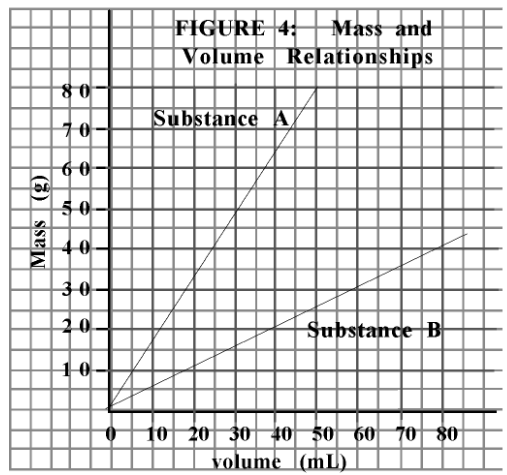
Substance B
Convert 0.23 kg to dg and write the answer in scientific notation.
2.3 x 103 dg
A metal cube has a mass of 250 grams and a density of 8.5 g/cm³. What is the volume of the metal cube?
29.41 cm3
 What kind of change is this? (Hint: What is the statue of liberty made of?)
What kind of change is this? (Hint: What is the statue of liberty made of?)
Chemical
mass= 150 g
Vinitial= 30 mL
Density= 10 g/mL
Find the final volume.
45 mL
If you have 40 mL of substance B, what would be the mass?
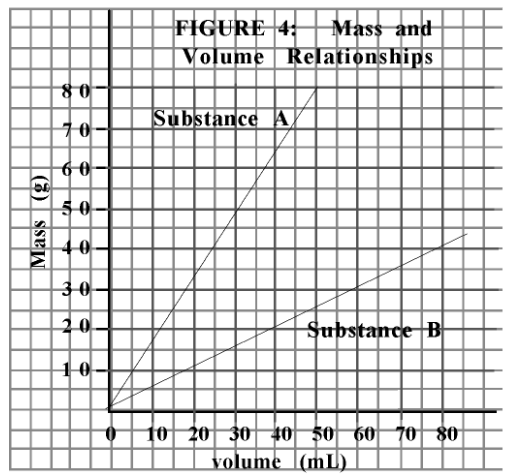
20 g
Which is the larger value? 6.98 g or 698mg
6.98 g
What is the mass of a cylinder of lead that is 2.5 cm in diameter, and 5.5 cm long? The density of lead is 11.4 g/mL and the volume of a cylinder is V=Πr2h
mass=307.78 grams