"101.3kPa and 298K"
Le Chatelier's
At 101.3 kPa and 298 K, a 1.0-mole sample of which compound releases the most energy when formed from gaseous elements.
NH3(g) or H2O(l) ?
H2O(l)
From Table I

2C(s) + H2(g) +227.4--> C2H2(g)
Why might this reaction be spontaneous?
There is an increase in entropy.
"Not lazy but crazy"
A system is at equilibrium. What is true about the rates of the forward and reverse reactions? What is true about the concentration of reactants and products?
REQUAL- Rates are EQUAL
CON CON- CONstant CONcentration
2CO(g) + O2(g) <=> 2CO2(g) delta H = –566.0
What happens to the system if these changes are made:
1. increase pressure
2. cool it down
They both will shift the equilibrium to the RIGHT. (more products made, and more heat given off).
H2(g) + I2(g) +53.0 -->2HI(g)
What happens to the reaction rate if the concentration of hydrogen gas is increased? Is decreased? Explain in terms of collision theory.
NOTE- Rate questions NOT Equilibrium questions
increased H2 will increase the reaction rate because there are more particles so more effective collisions
decreased H2 will decrease the reaction rate because there are fewer particles so fewer effective collisions
A reaction will occur if a collision occurs with what two properties?
Correct orientation and sufficient energy
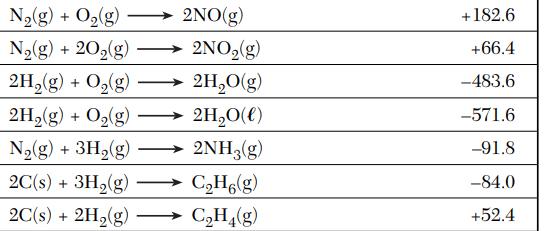
According to Table I, which equation represents a change resulting in the greatest quantity of energy released? Which has the greatest increase in entropy?
A) ![]()
B) ![]()
C) ![]()
Energy released: B) --> 2NH3
Entropy increased: A) --> C2H4(g)
Which type of solution demonstrates a solubility equilibrium?
Unsaturated, Saturated, Supersaturated
Saturated
2 SO2(g) + O2(g) ↔ 2 SO3(g) + heat
Which change will shift the equilibrium to the right?
A) increasing the temperature
B) increasing the pressure
C) decreasing the amount of SO2(g)
B) increasing the pressure
A system is at equilibrium. What is true about the rates of the forward and reverse reactions? What is true about the amounts of reactants and products?
Forward and reverse rates are EQUAL
Amounts of reactants and products are CONSTANT (not equal)
A 5.0-g sample of Zn and a 50.-milliliter sample of HCl are used in a chemical reaction. Which combination of these samples has the fastest reaction rate?
Zn strip, Zn powder, 1.0M HCl, 3.0M HCl
Zn powder
3.0M HCl

If each interval on the axis labeled "Potential Energy (kJ/mol)" represents 10. kJ/mol, what is the heat of reaction?
+30. kJ/mol
name each type of equilibrium
1) 2C(s) + 2H2(g) <=> C2H4(g)
2) NaCl (s) <=> Na+(aq) + Cl-(aq)
3) H2O(l) <=> H2O(g)
1) chemical equilibrium
2) solubility equilibrium
3) phase equilibrium
N2(g) + O2(g) + 182.6 kJ <=> 2NO(g)
When heat is added to the system, the concentration of N2(g) will_____ and the concentration of NO(g) will______.
N2(g) concentration will decrease
NO(g) concentration will increase
At 101.3 kPa and 298K, what is the sign of the enthalpy of reaction for:
2C(s) + 2H2(g) --> C2H4(g)
From Table I: +52.4kJ POSITIVE sign
2H₂(g) + O₂ (g) → 2H₂O (l)
If I increase the pressure on this reaction, what will happen to the rate? What happens if I decreases the pressure?
Increased pressure: The rate will increase- increasing the pressure on gaseous reactants causes more collisions, increasing the rate
Decreased pressure: The rate will decrease due to fewer collisions

What is the sign of the delta H for this reaction? Is it endothermic or exothermic?
The delta H will be negative. It is exothermic

At what time does the system reach equilibrium?
70 seconds
C(s) + O2(g) <--> CO2(g) + 393.5 kJ
How will the equilibrium change when the temperature of the system is increased?
The equilibrium will shirt to the LEFT
What is the entropy change for this reaction:
2C(s) + 2H2(g) --> C2H4(g)
Entropy increased. (s and g becoming g)
A catalyst increases the rate of a chemical reaction by providing an alternate reaction pathway that ...
has as lower activation energy

Which interval represents the activation energy of the reverse reaction? Which interval/s will be changed with the addition of a catalyst?
C is the reverse activation energy
B and C will be lowered with the addition of a catalyst
For a chemical system at equilibrium, the concentrations of both the reactants and the products must
A) decrease B) increase C) be constant D) be equal
C) be constant
N2(g) + 2O2(g) <=> 3NO2(g) +66.4kJ
List three ways to shift the equilibrium to the reactants.
1. increase the temperature
2. increase NO2
3. decrease N2 and/or O2

What is the heat of the activated complex?
What is the activation energy of the forward reaction?
heat of the activated complex = 50 kJ
activation energy of the forward reaction= 10 kJ