Define standard enthalpy of formation (ΔHf°)
The enthalpy change for the formation of 1 mole of a compound from its elements in their standard states
What is a bond enthalpy (bond energy)?
The average energy required to break one mole of a specified type of bond in gas-phase molecules, typically given in kJ/mol.
what is the triple point of water?
Where all three phases are present at the same time
What does ΔH°rxn represent for a balanced chemical equation?
The enthalpy change when the reaction as written (with its stoichiometric coefficients)
Define specific heat capacity (c). Include typical units.
Specific heat capacity is the amount of heat required to raise the temperature of 1 gram of a substance by 1 °C; units: J/(g·°C).
Given ΔHf°(CO2(g)) = -393.5 kJ/mol and ΔHf°(H2O(l)) = -285.8 kJ/mol, write the formation reaction for CO2(g) and for H2O(l).
- CO2(g): C(graphite) + O2(g) → CO2(g)
- H2O(l): 1/2 O2(g) + H2(g) → H2O(l)
CO2(g): -393.5kJ/mol
H2O(l): -285.8 kJ/mol
Estimate ΔH for CH4 + Cl2 → CH3Cl + HCl using average bond enthalpies: C–H = 413 kJ/mol,
Cl–Cl = 243 kJ/mol, C–Cl = 338 kJ/mol,
H–Cl = 431 kJ/mol. Show steps.
Bonds broken: one C–H (413) + Cl–Cl (243) = 656 kJ Bonds formed: one C–Cl (338) + one H–Cl (431) = 769 kJ ΔH ≈ 656 − 769 = −113 kJ
Label the main regions on a phase diagram (solid, liquid, gas)
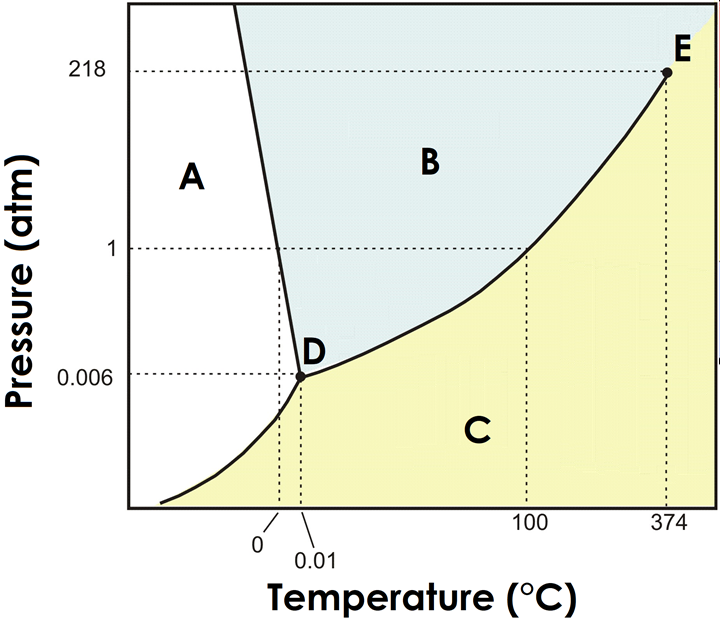
A= solid
B=liquid
C= gas
If the reaction A → B has ΔH = −120 kJ per mole of A, what is the enthalpy change when 2.5 mol of A reacts completely? Show the calculation.
ΔHtotal = (−120 kJ/mol) × 2.5 mol = −300 kJ
If energy is moves from the system to the surroundings, is the heat of the system positive or negative? explain why.
negative; heat was released
True or false: The ΔHf° of an element in its standard state is zero.
True
Use average bond enthalpies to estimate ΔH for
H2 + Cl2 → 2 HCl.
(H–H = 436 kJ/mol, Cl–Cl = 243 kJ/mol, H–Cl = 431 kJ/mol.)
ΔH= −183 kJ
On a heating curve for water, explain what happens to the temperature during a plateau (flat section) and why energy doesn't raise temperature at that segment.
Added heat energy does not raise the temperature because it is used exclusively to break intermolecular forces (potential energy) to change the state rather than increase molecular motion (kinetic energy).
The reaction: 2 NO(g) + O2(g) → 2 NO2(g) has ΔH° = −114 kJ
How much heat is released when 3.0 mol of NO2 is formed? Is this reaction endothermic or exothermic?
−171 kJ released; exothermic
Calculate the heat required to raise the temperature of 150.0 g of aluminum from 25.0 °C to 85.0 °C. Use c(Al) = 0.897 J/g·°C. Show formula and result.
q = m c ΔT
8073 J
Calculate ΔH°f for the reaction:
C(graphite) + 2 H2(g) → CH4(g)
using ΔHf° values: ΔHf°(CH4) = -74.8 kJ/mol,
ΔHf°(C(graphite)) = 0, ΔHf°(H2) = 0.
ΔH°rxn = −74.8 kJ/mol
Given: C(s) + O2 → CO2 ΔH = −393.5 kJ;
and 2 CO + O2 → 2 CO2 ΔH = −566.0 kJ.
Use Hess’s Law to find ΔH for: 2C(s) + O2 → 2 CO(g).
So ΔH = −110.5 kJ for 2 C + O2 → 2 CO
What is it called when a phase changed from a gas to a solid?
deposition
Given ΔH°f values: C6H12O6(s) = −1274 kJ/mol, CO2(g) = −393.5 kJ/mol, H2O(l) = −285.8 kJ/mol.
Calculate ΔH°rxn for:
C6H12O6(s) + 6 O2(g) → 6 CO2(g) + 6 H2O(l).
Products- Reactants
−2801.8 kJ
What does a coffee cup calorimeter measure?
The amount of heat energy transfer between two substances.
Use standard enthalpies of formation to calculate ΔH°rxn for:
4 NH3(g) + 3 O2(g) → 2 N2(g) + 6 H2O(l).
(Assume ΔHf°: NH3(g) = −45.9 kJ/mol; H2O(l) = −285.8 kJ/mol; elements = 0.)
ΔH°rxn= −765.6 kJ/mol
Provide a short step-by-step method (3–4 steps) for solving Hess’s Law problems when given multiple reactions with their ΔH values.
- Write target equation and list given equations with ΔH.
- Multiply or reverse given equations so their sum equals the target; when reversing change sign of ΔH; when multiplying scale ΔH by same factor.
- Add the adjusted equations and add the adjusted ΔH values.
- Cancel identical species that appear on both sides; result is ΔH for target reaction.
Which state is more dense for water, solid or liquid?
What direction will the solid/liquid equilibrium curve slant?
liquid; left
Use standard enthalpies of formation to calculate ΔH°rxn for: 2 NH3(g) + 1.5 O2(g) → N2(g) + 3 H2O(l).
(Assume ΔHf°: NH3(g) = −45.9 kJ/mol;
H2O(l) = −285.8 kJ/mol; elements = 0.)
−765.6 kJ
A 50.0 g sample of metal at 100.0 °C is dropped into 150.0 g of water at 22.0 °C. Final temperature is 26.0 °C. Assuming no heat loss and c(water) = 4.184 J/g·°C, calculate the specific heat capacity of the metal.
0.679 J/g·°C