Stored energy.
Potential Energy
Describe what happens to molecular movement as an object heats up.
Increase, molecules will move faster as heat is added.
At what position would there be the most potential energy?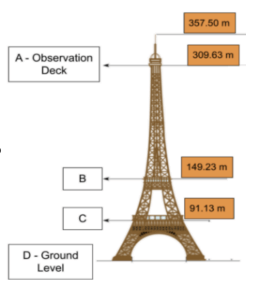
Position A (highest point)
What type of heat transfer is shown below? 
Conduction
What variable goes in A?
BONUS: Describe the relationship.
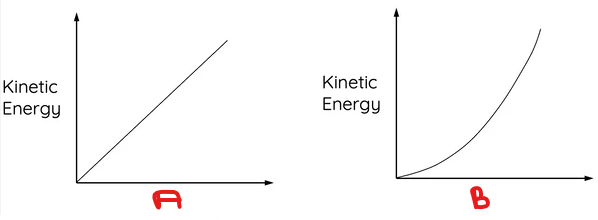
Mass.
Linear relationship.
Energy of motion.
Kinetic Energy
What happens to volume as you heat a substance?
Volume increases (molecules spread out).
What is the formula for PE?
BONUS 1: Name the variables.
BONUS 2: Name the units.
PE = mgh
m = mass
g= acceleration due to gravity
h = height
UNITS - Joules (J)
What is the relationship between temperature and the speed of molecules?
As the temperature increases, speed of the molecules increases (direct).
What variable goes in B?
BONUS: Describe the relationship.
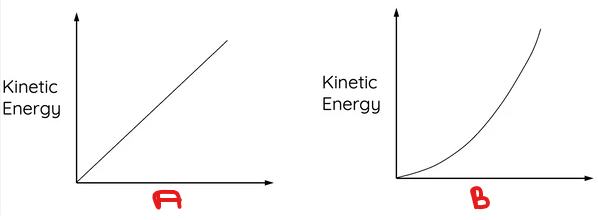
Velocity.
Exponential, nonlinear.

Potential energy

Conduction
What is the formula for KE?
BONUS 1: Name the variables.
BONUS 2: Name the units
KE = 1/2(m)(v2)
m = mass
v = velocity
UNITS - Joules (J)
Identify the heat transfer process shown below.
Convection
Describe the relationship between string length and the time to complete one swing.

As the string length increases, the time to complete one swing increases (direct relationship).
The sum of all KE and PE in a system.
Mechanical Energy
Why is heating something a physical change?
Heat will change the state (melting, boiling) or temperature without changing its composition.
What has more total energy?
A. A stationary ball on the ground
B. A stationary ball at the top of VMS
C. A rolling ball on the ground
D. A rolling ball at the top of VMS
D - a rolling ball at the top of VMS
High position = more PE, rolling/moving = more KE
What is thermal expansion?
When something is heated, molecules speed up and move farther apart, causing volume to increase.
Calculate the KE of a 8.5 kg bicycle moving at 7 m/s with correct units.
208.25 J

Kinetic energy

Radiation
The Empire State Building is 1,454 ft tall. The Eiffel Tower stands at 1,063 ft. Using quantitative data, explain how the amount of PE would change if an object was dropped from one of these buildings.
There would be more PE on top of the Empire State Building because it’s over 400 ft taller than the Eiffel Tower. The more height, the more PE is added to a system.
This type of heat transfer does not require a medium.
Radiation
Calculate the PE of an object with a mass of 8 kg at a height of 5 m above Earth's surface with correct units.
392.4 J