This type of bond forms between a metal and another metal
What is a metallic bond?
This is sodium fluoride (write the chemical formula)
What is NaF ?
That is CuF (write the chemical name)
What is copper (I) fluoride?
The prefixes in these covalent compounds determine what...?
What is the number of atoms?
All acids have this element bonded to them
What is hydrogen?
This chemical bond is formed between metals and nonmetals
What are ionic bonds?
CaBr2 is the chemical formula (write the chemical name)
What is calcium bromide?
That is TiO2 (write the chemical name)
What is titanium (IV) oxide?
This is N3H6 (write the chemical name)
What is trinitrogen hexahydride?
This is HBr (write the name of this STRONG ACID)
What is hydrobromic acid?
This chemical bond exists between two nonmetals such as H2
What is a covalent bond?
This is Mg3As2 (write the chemical name)
What is magnesium arsenide?
That is nickel (III) telluride (write the chemical formula)
What is Ni2S3 ?
This is CO3 (write the chemical name)
What is carbon trioxide?
This is H2SO4 (write the STRONG ACID name)
What is sulfuric acid?
A diagram of this type of bond could look like this: 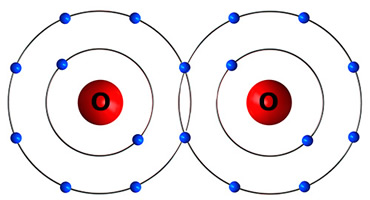
What is a covalent bond?
That is Ca3(PO4)2 (write the chemical name)
What is calcium phosphate?
That is CrP (write the chemical name)
What is chromium (III) phosphide?
This is sulfur tetroxide (write the chemical formula)
What is SO4 ?
That is HClO3 (write the acid name)
What is chloric acid?
A diagram of an ionic bond would look something like this...
(Example) 
This is beryllium nitrate (write the chemical formula)
What is Be(NO3)2
This is Mn2(SO3)5 (write the chemical name)
What is manganese (V) sulfite?
This is phosphorus decafluoride (write the chemical formula)
What is PF10 ?
All acids produce this ion in an aqueous (water-based) solution.
What is a hydrogen ion [H]+ / hydronium ion [H3O]+