
What describes calculating reactants and products?
Stoichiometry

What substance consists of a solute and solvent?
Solution
These are the starting substances in a chemical reaction.
Reactants
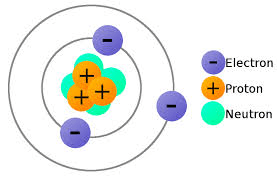
What is the number of electrons in an atom with 37 protons and 49 neutrons?
(1) 12
(2) 49
(3) 37
(4) 86
Remember:
Electrons = Protons
Neutrons = Atomic Mass - Electrons
37
Name one of the zones we follow in class
Silent Zone or Talk Zone

Which runs out first in a chemical reaction?
Limiting Reactant
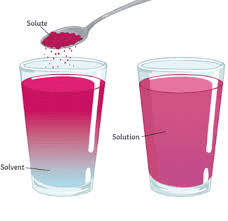
What is the substance that dissolves a solute?
Solvent

1 sandwich = 2 slices of bread + 1 slice cheese
You have:
6 slices bread
5 slices cheese
Bread
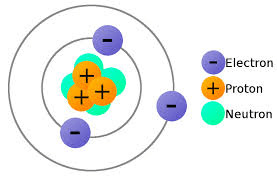
In the wave-mechanical model of the atom, electrons are located in regions called
(1) spectra
(2) quanta
(3) orbitals
(4) nuclei
Orbitals
Who is the most persevering student in class who still works when assignments get tough?

What is product yield compared to the ideal amount?
Percentage yield

A solution that holds as much solute as possible is _____.
Saturated

1 taco = 1 shell + 1 meat + 1 cheese
You have:
4 shells
6 meat
5 cheese
Which ingredient is the limiting reactant?
Shells

Which statement describes a chemical property of chlorine?
(1) Chlorine has an atomic radius of 100. pm.
(2) Chlorine reacts with sodium to form a salt.
(3) Chlorine has a melting point of 172 K.
(4) Chlorine is a green-yellow gas at STP.
Chlorine reacts with sodium to form a salt.
Who is the class instigator?
Koren

What do balancing numbers in equations create?
Mole ratio

What is a measure of solute is in a solution?
Concentration

Reaction:
2 H₂ + O₂ → 2 H₂O
You have:
6 H₂ molecules
2 O₂ molecules
Which reactant is the limiting reactant?
O2

Which substance can not be broken down by a chemical change?
(1) Fe
(2) FeSO4
(3) Fe2 O3
(4) Fe2 (SO4 ) 3
Fe
Name the three class trackers we utilize for chemistry class
Attendance
Homework
Participation

What is the mass of the product that is made in lab?
Actual yield

How many grams of salt in 100g of 5% salt solution?
5 grams

Reaction:
N₂ + 3 H₂ → 2 NH₃
You have:
2 N₂
3 H₂
Which reactant is limiting?
H2

When the pH value of a solution is changed from 4.0 to 7.0, what is the change in the concentration of hydronium ions?
(1) decreased by a factor of 1000
(2) increased by a factor of 1000
(3) decreased by a factor of 3
(4) increased by a factor of 3
Decreased by a factor of 1000
What is Mr. Paige's favorite anime?
A) Jujutsu Kaisen
B) Dragon Ball Z
C) Attack On Titan
D) My Hero Academia
Dragon Ball Z