What did John Dalton theorize about atoms of the SAME element?
They are the SAME
Which of the subatomic particles is the smallest?
The Electron
What are the electrons in the outermost orbital called?
Valence Electrons
Explain what an isotope is
An atom with the same number of protons but different number of neutrons compared to other atoms of the same element
What is the mass number of the atom shown in isotope notation below?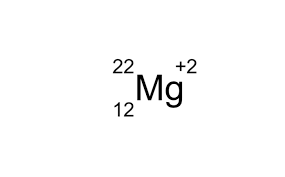
22
Who proposed the very FIRST atomic theory?
Democritus
Name the three subatomic particles that make up all atoms
Protons, Neutrons, Electrons
Can an orbital in a Bohr model hold less than the maximum amount of electrons allowed?
YES, but not more than the max amount
What two things does Atomic Number tell us about an atom?
The type of atom (element) and the number of protons
What is the atomic number of Tungsten?
74
Explain the nickname for J.J. Thomson's Model of the Atom
Plum Pudding Model - Positive pudding with negative plums floating around - DISCOVERED THE ELECTRON DJ
Where are all three subatomic particles located?
Protons - Nucleus
Neutrons - Nucleus
Electrons - Orbitals outside the Nucleus
What element does this Bohr Model represent?
Silicon
What is an orbital?
A region where there is a high probability of finding electrons
How many neutrons does the following isotope have?

10
How did Rutherford find the nucleus of the atom?
The Gold Foil Experiment
What two things do protons and neutrons have in common?
Which electron has more energy, the electrons that are closer to the nucleus, or the electrons that are further from the nucleus?
The electrons furthest from the nucleus have the most energy DJ
Explain what the Atomic Mass number tells us
The weighted average of the masses of all naturally occurring isotopes of an element
How many electrons does an atom of Mg+ have?
11 DJ
According to Bohr, how do electrons travel?
As WAVES - they do NOT move in predictable circles
How many electrons can fit in each of the first FOUR orbitals?
1st orbital - 2 electrons
2nd orbital - 8 electrons
3rd orbital - 8 electrons
4th orbital - 18 electrons
On your whiteboard, draw the Bohr Model for Boron

Explain the difference between a cation and an anion
Cation - positively charged atom/molecule that has LOST an electron
Anion - negatively charged atom/molecule that has GAINED an electron
How many protons are in an atom of Silver 2+?
47