Salt and pepper is this type of mixture
Heterogeneous
Soluble or insoluble in water: CaCO3
Insoluble
If 4.51 grams of NaCl are dissolved in 125 grams of water, what is the percent by mass?
3.47%
What stress affects the equilibrium constant
temperature/heat
The formula for permanganic acid
HMnO4
pH is a logarithmic scale that ranges from __ to __.
0 to 14
The thing that does the dissolving in a solution
Solvent
A __________ solution contains the maximum amount of solute for a given amount of solvent.
Saturated
If 23.0 grams of NaOH are dissolved in 435 mL of water, what is the molarity of the solution?
1.32 M NaOH
Write the equilibrium expression for
2HBr(g) <-> H2(g) + Br2(l)
Keq = [H2]/[HBr]2
A solution has a pH of 3.25, what is the hydronium ion concentration?
5.62 x 10-4 M
What is equal when a reaction is at equilibrium?
Forward and reverse reaction rates.
Increasing the temperature would ________ the solubility of solids and _________ the solubility of gasses.
Solids - Increase
Gasses - Decrease
What mass of KClO3 will be needed to make a saturated solution in 100 grams of water at 30 C?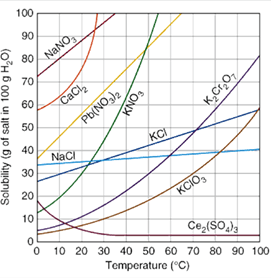
10 grams
If you created a 10.0 M solution of sulfuric acid yesterday, what would you have put on the label for the stock solution?
10.0 M
H2SO4
5/5/21
Your Name
For the reaction
2NO(g) + Br2(g) <--> 2NOBr(g)
the concentrations at equilibrium are as follows:
[NO] = 0.50 M [Br2] = 0.25 M [NOBr] = 3.5 M
Calculate Keq
196
What is the pH of a solution with a hydroxide concentration of 4.76 x 10-3?
pH = 11.68
Substances like water that can act as both an acid and a base are referred to as being ________.
Amphoteric
Would liquid methane typically be soluble in water?
No, it is nonpolar and like dissolves like
The three methods by which the rate of dissolution can be changed.
Agitation, temperature, surface area
What volume of a 12.4 M stock solution of NaOH would you need in order to create 200.0 mL of a .100 M NaOH solution? (Answer in mL)
1.61 mL
2CO (g) + O2 (g) → 2CO2 (g) + 566.0 kJ
What shift would occur if pressure were decreased?
Towards the left
Identify the acid, base, conjugate acid, and conjugate base in the following equation:
HCO3- + HNO3 --> H2CO3 + NO3-
Acid - HNO3
Base - HCO3-
Conj. Acid - H2CO3
Conj. Base - NO3-
A strong acid is one that will ionize _________, and is represented in equations with a ________ arrow.
100%, -->
What type of solution would get when you combine an acid and base?
Salt water (salt + water)
Which of the following would have the greatest effect on shifting the boiling point and freezing point of water? Sugar, NaCl, NaOH, HCl, Ba(OH)2, Na3PO4
Na3PO4
It will produce the most ions in solution (4). Remember that colligative properties depend only on the number of particles, not the particle identity.
Potassium hydroxide reacts with sulfuric acid to produce potassium sulfate and water. What mass of potassium hydroxide would be required to completely react with 50.0 mL of a 4.32 M solution of acid?
24.2 grams KOH
Keq for the following reaction is 15:
H2(g) + Cl2(g) <--> 2HCl(g)
At some other point the concentrations are as follows:
[H2] = .42 M [Cl2] = .075 M [HCl] = .95 M
Calculate Q and using Keq, determine the direction the reaction will proceed to reach equilibrium.
Q = 29
Proceed left towards the reactants
32.0 mL of HCl is neutralized with 52.3 mL of .500 M NaOH. What is the concentration of HCl?
.817 M HCl
Which solution below would have the greatest effect on lowering the freezing point?
2.0 M NaOH
1.5 M MgCl2
1.5 M K3PO4
5.0 M C6H12O6
1.5 M K3PO4