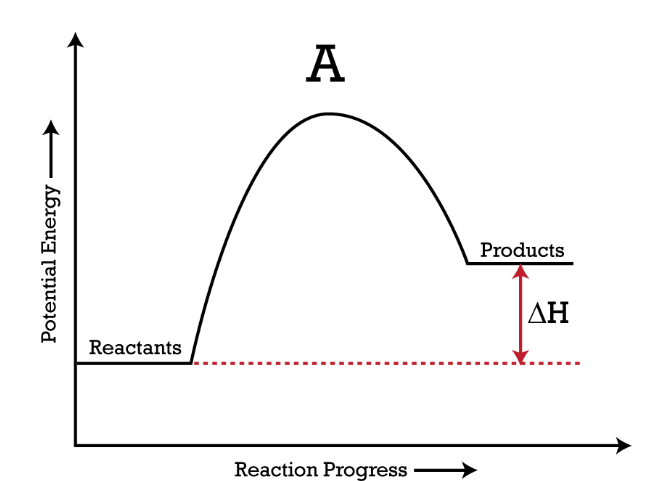
 Based on the picture, where is the energy going?
Based on the picture, where is the energy going?
(Entering/Leaving) the system
Leaving the System
Based on the chart below that lists the specific heat, which of the following object will experience the least temperature change?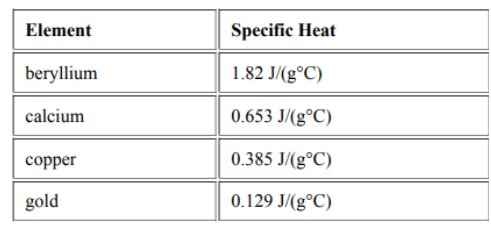
Beryllium
This is the heat content of a system at constant temperature and it is denoted as "ΔH "
How does heat spontaneously move from one object to another?
Heat moves from a hotter object to a cooler object.
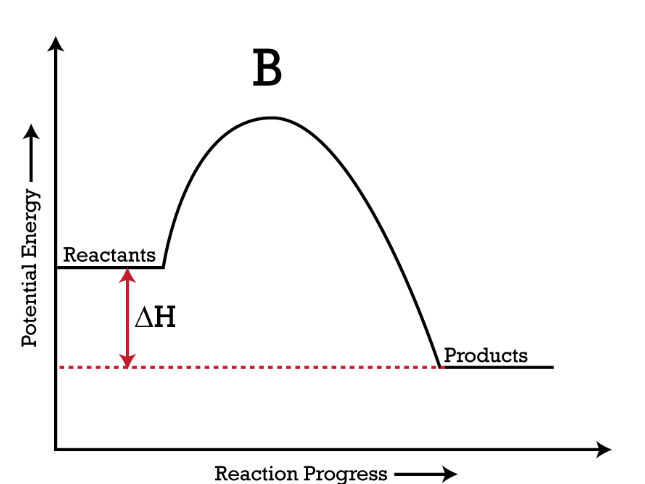
Based on the picture, where is the energy going?
(Entering/Leaving) the system
Entering the System
Rank the following objects from least to greatest regarding their temperature change when heated.
(Least Temperature Change --> Greatest Temperature Change)
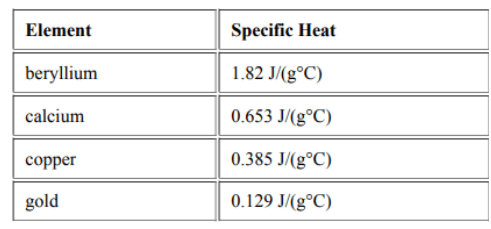
beryllium < calcium <copper < gold
This is a measure of the number of possible ways that the energy of a system can be distributed due to the randomness/disorder of particles in a system.
entropy
Determine the number of moles of 6.08 grams of SiO2.
(Hint: You need to use the periodic table for this to get the molar mass)
0.10 moles
Molar Mass: 60.08 g/mol
Based on the thermochemical equation, what kind of reaction will this be?

exothermic
Explain the meaning of this equation relating to calorimetry:
Qwater = -Qmetal
The heat/energy lost by the metal is transferred to the water. Energy is conserved in the process.
What are the two units of energy that we use for this unit?
Joules (SI Unit)
Calories (heat required to raise the temperature of one gram of water through 1 degree Celsius)
Please determine the standard enthalpy (ΔH) for this reaction:

102.23 kJ
Based on the thermochemical equation, what kind of reaction will this be?

Endothermic
How much energy being absorbed in Joules is required to heat 50.0 grams of iron with a temperature change of 55°C? The specific heat of iron is 0.444 J/g°C.
Useful Equations
q = mcΔT
Answer is 1220 J
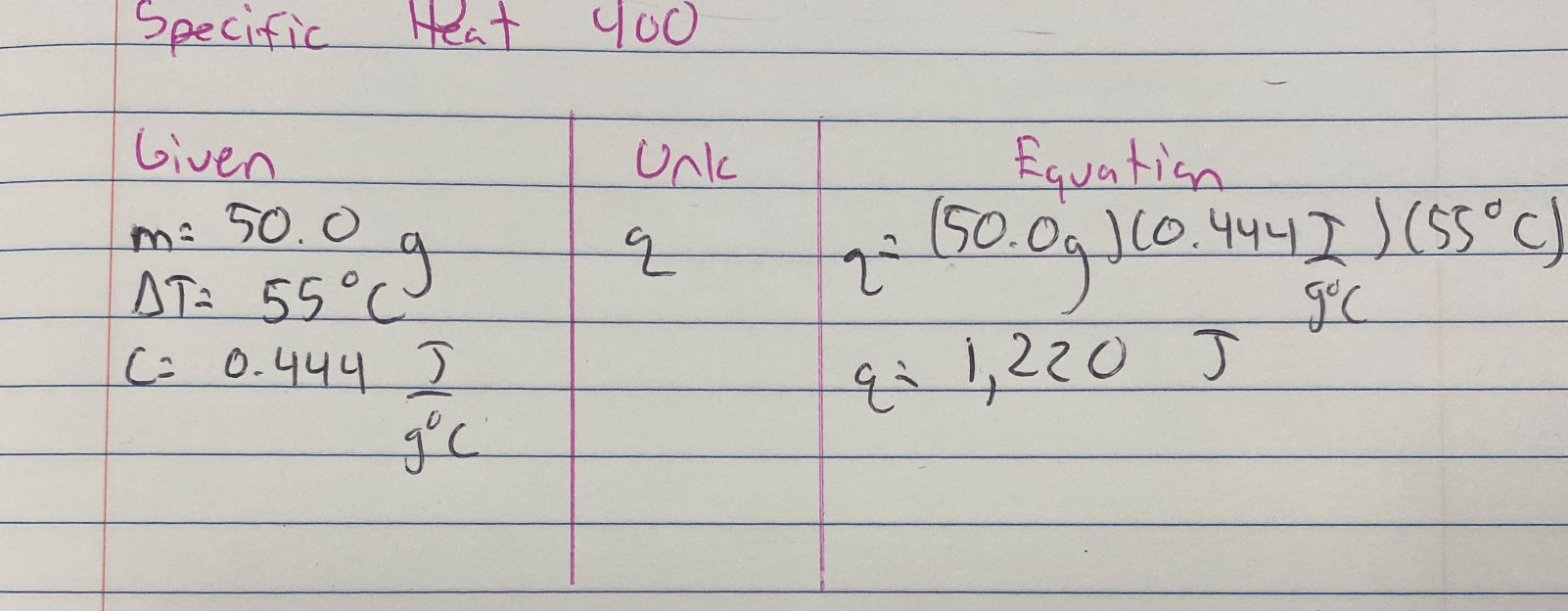
Explain the difference between endothermic and exothermic:
Endothermic - Energy is being absorbed in the reaction, heat is going into the system
Exothermic - Energy is being released from the reaction, heat is going out of the system
Calculate the Standard Heat of Formation (ΔH) for this reaction. Is this exothermic or endothermic?
SO3 (g) + H2O (g) --> H2SO4 (aq)
Given that the ΔH of:
H2SO4: -909.27 kJ
H2O: -241.8 kJ
SO3: -395.7 kJ
Answer: -271.77 kJ (Exothermic)
Work:
H2SO4 ---> H2O + SO3
(-909.27) - (-241.8 + -395.7) = -271.77 kJ
Please give one real life example of an exothermic and endothermic reaction
Answers might vary based on Teacher's discretion
How much energy is released when 25.0 grams of aluminum metal is cooled from 125°C to 35°C? The specific heat of aluminum is 0.900 J/g°C.
Useful Equations
q = mc(Tfinal - Tinitial)
Answer is -2030 J
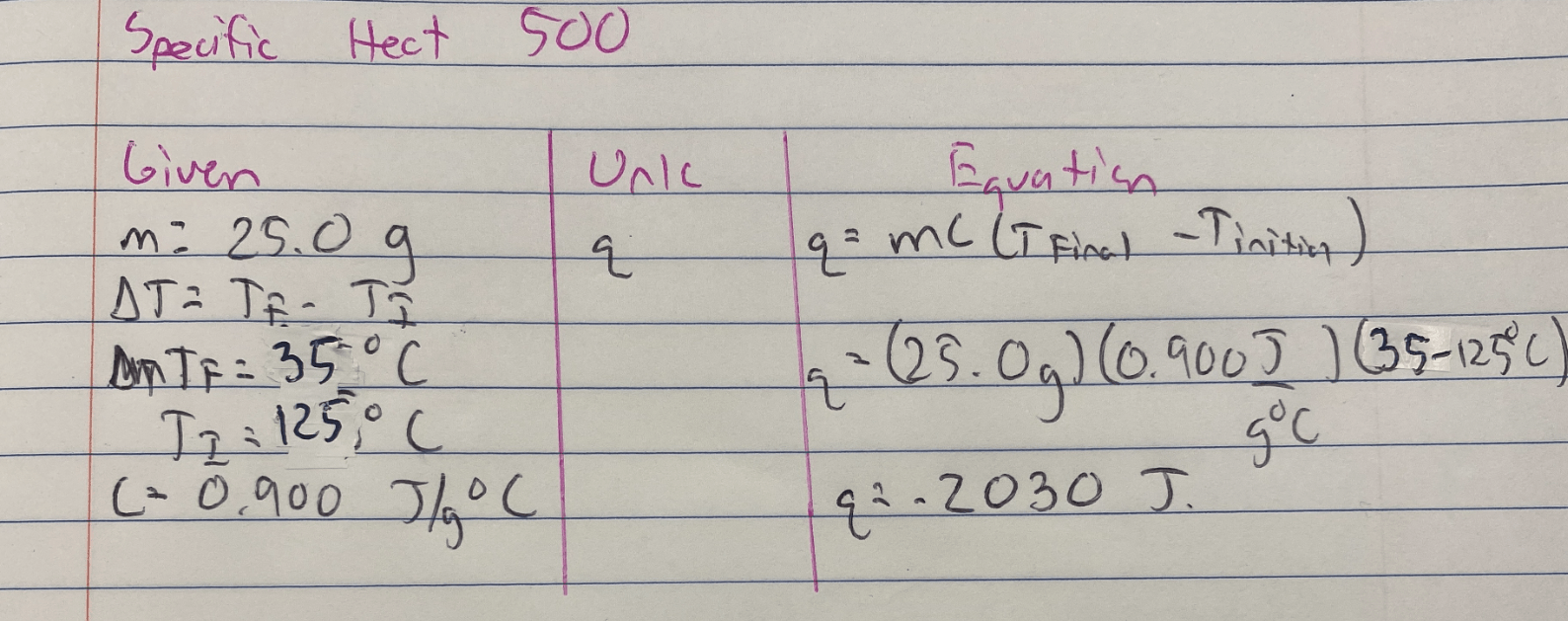
When we talk about the perspective about the heat exchange that is happening in the universe, what two things that it consists of?
System and Surrounding

Game Plan: Flip Equation # 1 Only
ΔH = 72.5 kJ