The one or two letters used to represent an element
What is the chemical symbol?
The charge on the fluorine ion
What is -1?
The kinds of atoms covalent bonds form between
What are non-metals?
The reaction classification for the formation of table salt from Na and Cl
Synthesis
The number of valence electrons for oxygen
The group number for Magnesium (Mg)
What is group 2?
The number of electrons lost/gained for the Strontium (Sr) ion
What is 2 electrons lost?
Number of total electrons represented in the lewis dot structure for CO2
What is 16 electrons?
The reason atoms must be balanced on both sides of a chemical reaction
What is law of conservation of mass?
The number of total orbitals in one D subshell
What is 5 orbitals?
The atomic mass number for Indium (In)
What is 114.82 amu?
The chemical formula of a compound between aluminum and oxygen
What is Al2O3?
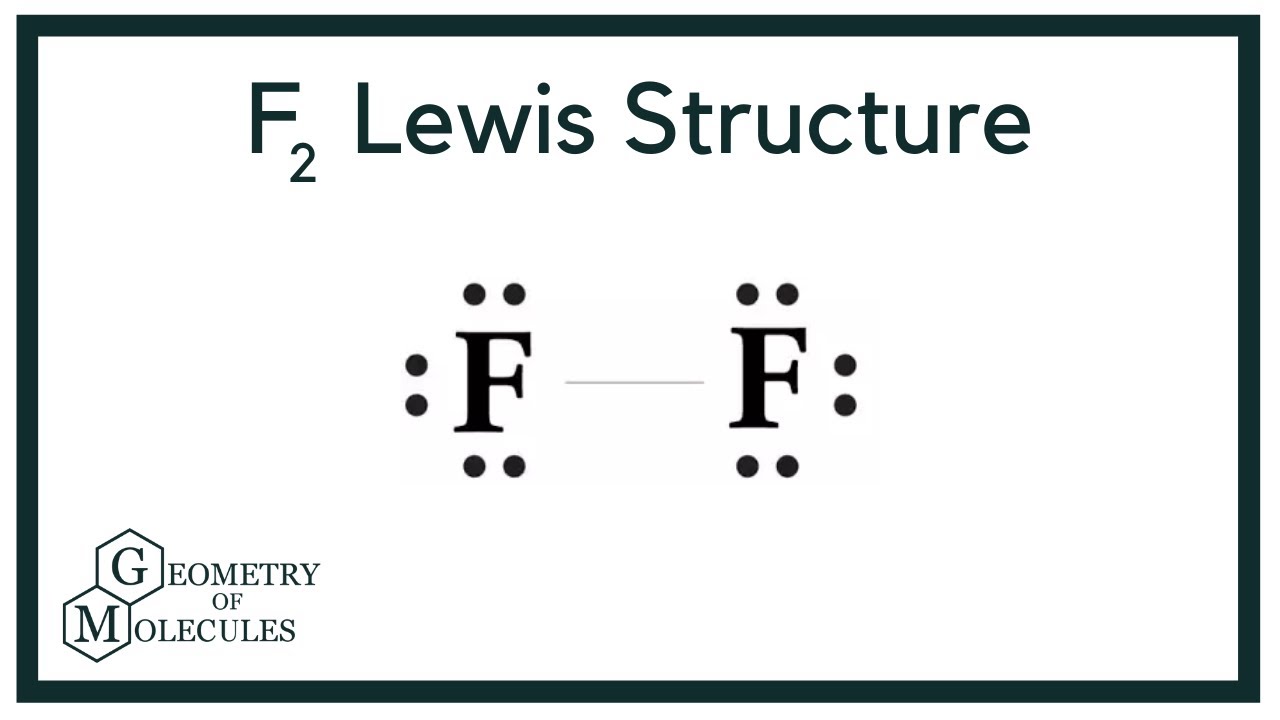
Lewis dot structure of F2
Which one is indication that a chemical reaction has occurred?
A. Change in mass
B. Change in color
C. Change in density
D. Change in state of matter
What is B/color?
What is 1s22s22p63s23p5 OR [Ne]3s23p5
The approximate number of neutrons in Chlorine (atomic mass of 35.45 amu)
What is 17-18 neutrons?
The name of a positive ion
What is a cation?
The number of covalent bonds a Carbon (C) atom will form
What is 4?
The reaction classification for:
4Fe + 3O2 --> 2Fe2O3
What is synthesis?
The electron configuration of the Selenium (Se) ion
What is 1s22s22p63s23p64s23d104p4 OR [Ar]4s23d104p4?
The mass of electrons in Rubidium (atomic mass of 85.47 amu)
What is 0?
The conductivity of ionic solids
What is not conductive?
Lewis dot structure of water
(we will draw)
The coefficient on oxygen gas in the combustion of methane (CH4)
What is 2?
The short form electron configuration for Astatine (At)
What is [Xe]6s25d104f146p5