Name the 3 states of matter
Solid, Liquid, Gas
When a liquid turns to a solid it is called
Melting
Define a mixture
2 or more substances, not chemically bonded together
Name a soluble substance
salt, sugar, etc
Name this piece of equipment
Conical Flask
Which state is this?
liquid
When a liquid turns to a gas it is called
evaporating
Define an element
made of only one type of atom
What is distillation used for?
removing a solvent from a mixture
Name this piece of equipment
round - bottom flask
How are the particles arranged in a solid?
Neat, organised rows, particles touching eachother
When a gas turns to a liquid it is called
condensation
How would you separate a liquid from an insoluble solid?
Using filtration
What piece of equipment would you use to find 25ml of water?
a measuring cylinder
Which state has the highest amount of energy?
Gas
when a solid turns to a gas it is called
sublimation
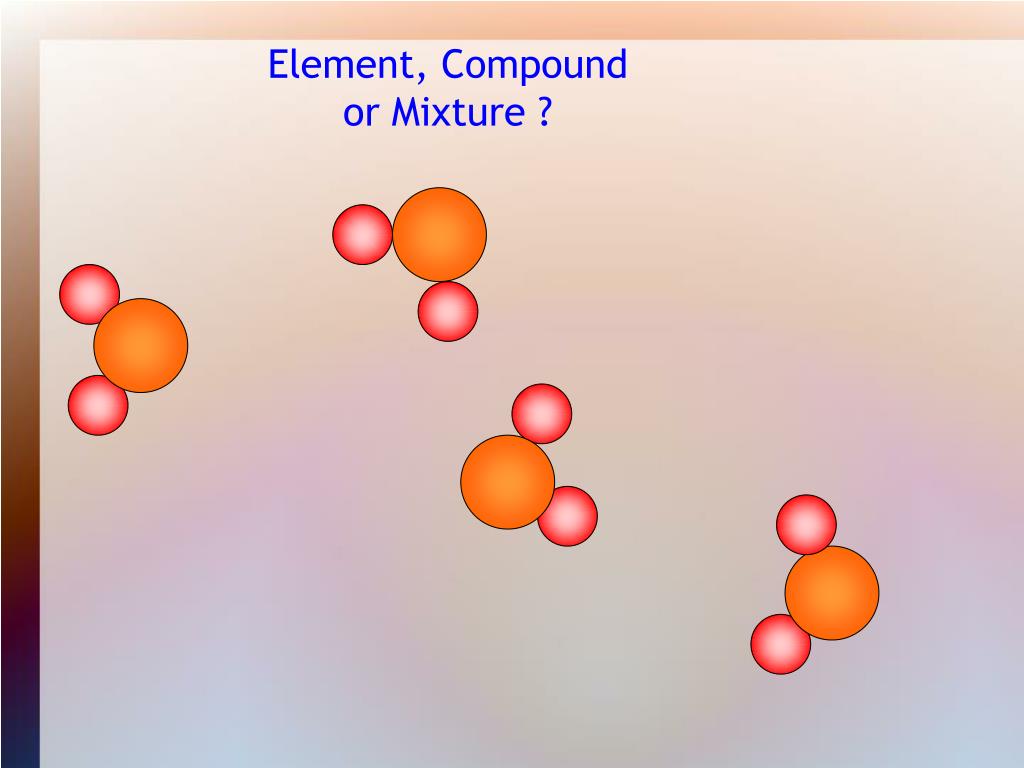
compound
How would you separate 2 soluble substances?
chromatography

Name part D
Filtrate
What is the difference between particles in a solid and a liquid
what happens to the particles when a solid is melted.
They gain energy and begin moving more
explain whether you can have a pure mixture or a pure compound
you can have a pure compound, but not a pure mixture
Pure = made up of only 1 type of substance
what factors can affect solubility?
temperature
What is this equipment set up being used for?

distillation